Preferential mobilisation of oxidised iron by slab-derived hydrous silicate melts
Affiliations | Corresponding Author | Cite as | Funding information- Share this article




-
Article views:184Cumulative count of HTML views and PDF downloads.
- Download Citation
- Rights & Permissions
top
Abstract
Figures and Tables
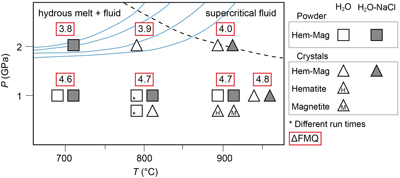 Figure 1 Pressure and temperature conditions of experimental runs employing powders (squares) or single crystals (triangles) as starting materials. The oxygen fugacity is reported relative to the Fayalite-Magnetite-Quartz (FMQ) buffer. The dotted line denotes the critical curve in the haplogranite–H2O system (Bureau and Keppler, 1999). Solid lines represent hot subduction geothermal paths (Syracuse et al., 2010). | 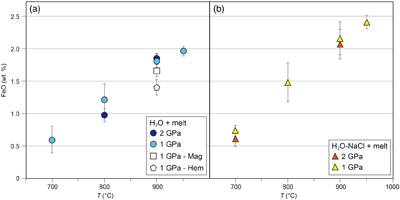 Figure 2 Experimental FeO contents (wt. %) measured by EMPA in the quenched glasses, as a function of temperature for (a) the H2O + melt system and (b) the H2O-NaCl + melt system. | 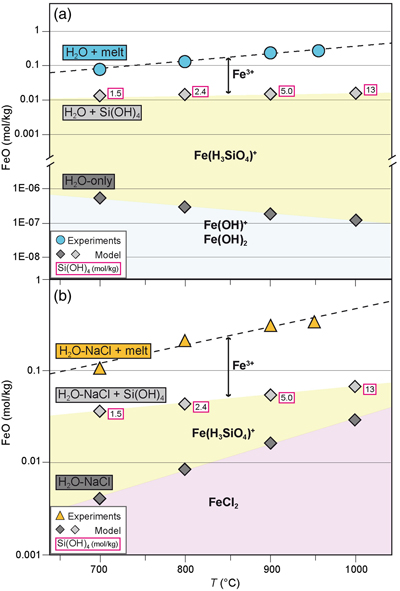 Figure 3 Calculated hematite–magnetite solubility (DEW model, Sverjensky et al., 2014) in (a) aqueous and (b) saline fluids at 1 GPa, and comparison with experimental results in silicate melts. Dark grey diamonds: FeO content (mol/kg) in the H2O-only and H2O-NaCl system. Light grey diamonds: FeO (mol/kg) in Si(OH)4-bearing fluids (mol/kg, pink boxes). Circles (H2O-only) and triangles (H2O-NaCl) denote experimental FeO contents in hydrous silicate melts (mol/kg) from this study. Error bars are smaller than the symbol size. The black double-ended arrow shows the effect of oxidised iron. Coloured fields denote the dominance of the indicated solute species in the fluid. | 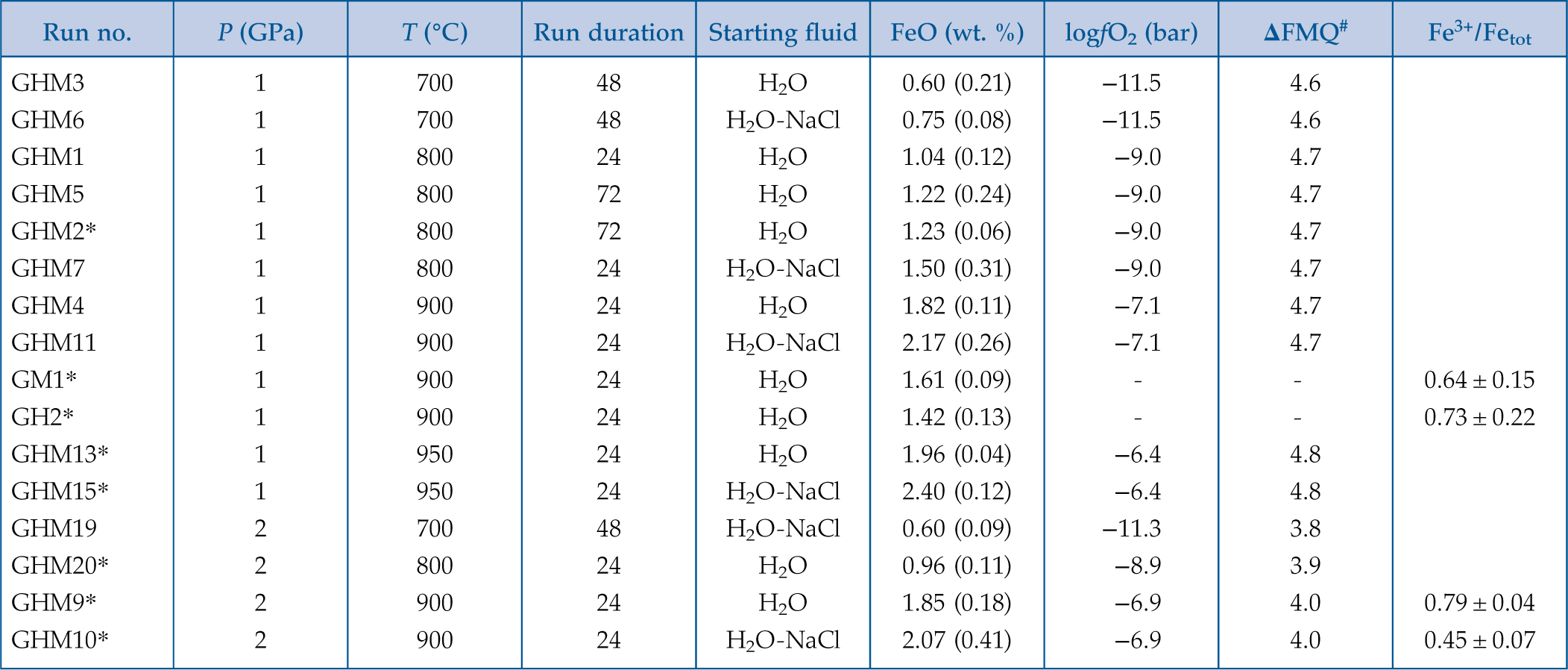 Table 1 Experimental run conditions, FeO content (in wt. %) and Fe3+/Fetot ratios in the quenched haplogranite glasses as determined by EMPA and Mössbauer spectroscopy, respectively. |
| Figure 1 | Figure 2 | Figure 3 | Table 1 |
top
Introduction
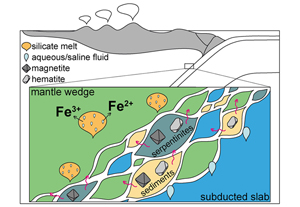
Arc magmas are characterised by a higher oxidation degree than magmas from other tectonic settings, including mid-oceanic ridge and ocean arc basalts (Carmichael, 1991
Carmichael, I.S.E. (1991) The redox states of basic and silicic magmas: a reflection of their source regions? Contributions to Mineralogy and Petrology 106, 129–141. https://doi.org/10.1007/BF00306429
; Kelley and Cottrell, 2009Kelley, K.A., Cottrell, E. (2009) Water and the oxidation state of subduction zone magmas. Science 325, 605–607. https://doi.org/10.1126/science.1174156
; Brounce et al., 2014Brounce, M.N., Kelley, K.A., Cottrell, E. (2014) Variations in Fe3+/∑Fe of Mariana Arc Basalts and Mantle Wedge fO2. Journal of Petrology 55, 2514–2536. https://doi.org/10.1093/petrology/egu065
). The oxidised signature is thought to arise from the influx of oxidising slab-derived fluids or melts into the mantle wedge (e.g., Burgisser and Scaillet, 2007Burgisser, A., Scaillet, B. (2007) Redox evolution of a degassing magma rising to the surface. Nature 445, 194–197. https://doi.org/10.1038/nature05509
; Debret and Sverjensky, 2017Debret, B., Sverjensky, D.A. (2017) Highly oxidising fluids generated during serpentinite breakdown in subduction zones. Scientific Reports 7, 1–6. https://doi.org/10.1038/s41598-017-09626-y
; Bénard et al., 2018Bénard, A., Klimm, K., Woodland, A.B., Arculus, R.J., Wilke, M., Botcharnikov, R.E., Shimizu, N., Nebel, O., Rivard, C., Ionov, D.A. (2018) Oxidising agents in sub-arc mantle melts link slab devolatilisation and arc magmas. Nature Communications 9, 1–10. https://doi.org/10.1038/s41467-018-05804-2
; Walters et al., 2020Walters, J.B., Cruz-Uribe, A.M., Marschall, H.R. (2020) Sulfur loss from subducted altered oceanic crust and implications for mantle oxidation. Geochemical Perspectives Letters 13, 36–41. https://doi.org/10.7185/geochemlet.2011
; Evans and Frost, 2021Evans, K.A., Frost, B.R. (2021) Deserpentinization in subduction zones as a source of oxidation in arcs: A reality check. Journal of Petrology 62, 1–32. https://doi.org/10.1093/petrology/egab016
), although the actual oxidising agent and the transport mechanism remain a topic of discussion (e.g., Piccoli et al., 2019Piccoli, F., Hermann, J., Pettke, T., Connolly, J.A.D., Kempf, E.D., Vieira Duarte, J.F. (2019) Subducting serpentinites release reduced, not oxidized, aqueous fluids. Scientific Reports 9, 1–7. https://doi.org/10.1038/s41598-019-55944-8
; Tollan and Hermann, 2019Tollan, P., Hermann, J. (2019) Arc magmas oxidized by water dissociation and hydrogen incorporation in orthopyroxene. Nature Geoscience 12, 667–671. https://doi.org/10.1038/s41561-019-0411-x
). Iron, as the most abundant redox-sensitive element in the upper mantle, may play a pivotal role in these processes, and constraints on its mobility and speciation in slab-derived phases are thus critical to elucidating the range of redox state conditions of subduction zones (Frost and McCammon, 2008Frost, D.J., McCammon, C.A. (2008) The Redox State of Earth’s Mantle. Annual Review of Earth and Planetary Sciences 36, 389–420. https://doi.org/10.1146/annurev.earth.36.031207.124322
; Kelley and Cottrell, 2009Kelley, K.A., Cottrell, E. (2009) Water and the oxidation state of subduction zone magmas. Science 325, 605–607. https://doi.org/10.1126/science.1174156
; Evans, 2012Evans, K.A. (2012) The redox budget of subduction zones. Earth-Science Reviews 113, 11–32. https://doi.org/10.1016/j.earscirev.2012.03.003
).Iron oxides such as hematite, Fe2O3, and magnetite, Fe3O4, are by-products of serpentinisation (e.g., Carbonin et al., 2015
Carbonin, S., Martin, S., Tumiati, S., Rossetti, P. (2015) Magnetite from the Cogne serpentinites (Piemonte ophiolite nappe, Italy). Insights into seafloor fluid–rock interaction. European Journal of Mineralogy 27, 31–50. https://doi.org/10.1127/ejm/2014/0026-2410
; Debret et al., 2015Debret, B., Bolfan-Casanova, N., Padron-Navarta, J.A., Martin-Hernandez, F., Andreani, M., Garrido, C.J., Sánchez-Vizcaíno V.L., Gómez-Pugnaire M.T., Mu˜noz, M., Trcera, N. (2015) Redox state of iron during high-pressure serpentinite dehydration. Contributions to Mineralogy and Petrology 169, 1–18. https://doi.org/10.1007/s00410-015-1130-y
), and they have been identified in seafloor metasediments (e.g., Ague et al., 2022Ague, J.J., Tassara, S., Holycross, M.E., Li, J.-L., Cottrell, E., Schwarzenbach, E.M., Fassoulas, C., John, T. (2022) Slab-derived devolatilization fluids oxidized by subducted metasedimentary rocks. Nature Geoscience 15, 320–326. https://doi.org/10.1038/s41561-022-00904-7
). Consequently, their solubility may control the iron budget in slab-derived fluids and melts. To date, most experimental studies on the solubility of hematite and/or magnetite in aqueous fluids (Chou and Eugster, 1977Chou, I.M., Eugster, H.P. (1977) Solubility of magnetite in supercritical chloride solutions. American Journal of Science 277, 1296–1314. https://doi.org/10.2475/ajs.277.10.1296
; Boctor et al., 1980Boctor, N.Z., Popp, R.K., Frantz, J.D. (1980) Mineral-solution equilibria—IV. Solubilities and the thermodynamic properties of FeCl20 in the system Fe2O3-H2-H2O-HCl. Geochimica et Cosmochimica Acta 44, 1509–1518. https://doi.org/10.1016/0016-7037(80)90115-5
; Frantz et al., 1981Frantz, J.D., Popp, R.K., Boctor, N.Z. (1981) Mineral-solution equilibria-V. Solubilities of rock-forming minerals in supercritical fluids. Geochimica et Cosmochimica Acta 45, 69–77. https://doi.org/10.1016/0016-7037(81)90264-7
; Scholten et al., 2019Scholten, L., Schmidt, C., Lecumberri-Sanchez, P., Newville, M., Lanzirotti, A., Sirbescu, M.L.C., Steele-MacInnis, M. (2019) Solubility and speciation of iron in hydrothermal fluids. Geochimica et Cosmochimica Acta 252, 126–143. https://doi.org/10.1016/j.gca.2019.03.001
) and melts (Simon et al., 2004Simon, A.C., Pettke, T., Candela, P.A., Piccoli, P.M., Heinrich, C.A. (2004) Magnetite solubility and iron transport in magmatic-hydrothermal environments. Geochimica et Cosmochimica Acta 68, 4905–4914. https://doi.org/10.1016/j.gca.2004.05.033
; Bell and Webster, 2015Bell, A.S., Webster, J.D. (2015) Dissolved Cl, oxygen fugacity, and their effects on Fe behavior in a hydrous rhyodacitic melt. American Mineralogist 100, 1595–1599. https://doi.org/10.2138/am-2015-5197
), have been conducted at moderate pressures, less than 0.4 GPa, that are not relevant for sub-arc settings. A single study of hematite solubility in H2O-NaCl fluid at 1 GPa and 800 °C by Wykes et al. (2008)Wykes, J.L., Newton, R.C., Manning, C.E. (2008) Solubility of andradite, Ca3Fe2Si3O12, in a 10 mol% NaCl solution at 800 °C and 10 kbar: Implications for the metasomatic origin of grandite garnet in calc-silicate granulites. American Mineralogist 93, 886–892. https://doi.org/10.2138/am.2008.2736
reported dissolved iron contents below 0.01 mol/kg. This value implies Fe3+ fluxes of 109 mol/year, which are three to four orders of magnitude lower than the estimated redox budget flux (55 ± 13 × 1012 mol/year; Evans, 2012Evans, K.A. (2012) The redox budget of subduction zones. Earth-Science Reviews 113, 11–32. https://doi.org/10.1016/j.earscirev.2012.03.003
). Alternatively, silica-bearing melt-like fluids have been suggested as an efficient agent for the transfer of oxidised iron (Parkinson and Arculus, 1999Parkinson, I.J., Arculus, R.J. (1999) The redox state of subduction zones: Insights from arc-peridotites. Chemical Geology 160, 409–423. https://doi.org/10.1016/S0009-2541(99)00110-2
; Hermann et al., 2006Hermann, J., Spandler, C., Hack, A., Korsakov, A.V. (2006) Aqueous fluids and hydrous melts in high-pressure and ultra-high pressure rocks: Implications for element transfer in subduction zones. Lithos 92, 399–417. https://doi.org/10.1016/j.lithos.2006.03.055
; Kelley and Cottrell, 2009Kelley, K.A., Cottrell, E. (2009) Water and the oxidation state of subduction zone magmas. Science 325, 605–607. https://doi.org/10.1126/science.1174156
; Bénard et al., 2018Bénard, A., Klimm, K., Woodland, A.B., Arculus, R.J., Wilke, M., Botcharnikov, R.E., Shimizu, N., Nebel, O., Rivard, C., Ionov, D.A. (2018) Oxidising agents in sub-arc mantle melts link slab devolatilisation and arc magmas. Nature Communications 9, 1–10. https://doi.org/10.1038/s41467-018-05804-2
) owing to its low solubility in aqueous phases (Wykes et al., 2008Wykes, J.L., Newton, R.C., Manning, C.E. (2008) Solubility of andradite, Ca3Fe2Si3O12, in a 10 mol% NaCl solution at 800 °C and 10 kbar: Implications for the metasomatic origin of grandite garnet in calc-silicate granulites. American Mineralogist 93, 886–892. https://doi.org/10.2138/am.2008.2736
). Yet, the capacity of hydrous silicate melts to transport oxidised iron at sub-arc conditions remains unconstrained.Here, we present experimental data on the solubility of hematite–magnetite assemblages in hydrous haplogranitic melts, which mimic melts produced by partial melting of subducted metasediments, to constrain their ability to transfer oxidised iron at sub-arc conditions. Combined with Mössbauer spectroscopy analysis of the recovered glasses and thermodynamic modelling, we demonstrate the higher capacity of hydrous silicate melts to mobilise oxidised iron compared to aqueous and chlorinated fluids at sub-arc conditions. The dissolution of iron oxides in slab-derived silicate melts thus provides a viable mechanism for the transfer of oxidised iron from the slab to the mantle wedge and to explain the oxidised nature of arc magmas (Kelley and Cottrell, 2009
Kelley, K.A., Cottrell, E. (2009) Water and the oxidation state of subduction zone magmas. Science 325, 605–607. https://doi.org/10.1126/science.1174156
).top
Solubility and Redox State of Iron in Hydrous Silicate Melts
A total of 16 hematite–magnetite solubility experiments were performed at 1–2 GPa and 700–950 °C (Fig. 1, Table 1, Fig. S-1, and Table S-1). Experimental and analytical details can be found in the Supplementary Information.

Figure 1 Pressure and temperature conditions of experimental runs employing powders (squares) or single crystals (triangles) as starting materials. The oxygen fugacity is reported relative to the Fayalite-Magnetite-Quartz (FMQ) buffer. The dotted line denotes the critical curve in the haplogranite–H2O system (Bureau and Keppler, 1999
Bureau, H., Keppler, H. (1999) Complete miscibility between silicate melts and hydrous fluids in the upper mantle: Experimental evidence and geochemical implications. Earth and Planetary Science Letters 165, 187–196. https://doi.org/10.1016/S0012-821X(98)00266-0
). Solid lines represent hot subduction geothermal paths (Syracuse et al., 2010Syracuse, E.M., van Keken, P.E., Abers, G.A. (2010) The global range of subduction zone thermal models. Physics of the Earth and Planetary Interiors 183, 73–90. https://doi.org/10.1016/j.pepi.2010.02.004
).Table 1 Experimental run conditions, FeO content (in wt. %) and Fe3+/Fetot ratios in the quenched haplogranite glasses as determined by EMPA and Mössbauer spectroscopy, respectively.
| Run no. | P (GPa) | T (°C) | Run duration | Starting fluid | FeO (wt. %) | logfO2 (bar) | ΔFMQ# | Fe3+/Fetot |
| GHM3 | 1 | 700 | 48 | H2O | 0.60 (0.21) | −11.5 | 4.6 | |
| GHM6 | 1 | 700 | 48 | H2O-NaCl | 0.75 (0.08) | −11.5 | 4.6 | |
| GHM1 | 1 | 800 | 24 | H2O | 1.04 (0.12) | −9.0 | 4.7 | |
| GHM5 | 1 | 800 | 72 | H2O | 1.22 (0.24) | −9.0 | 4.7 | |
| GHM2* | 1 | 800 | 72 | H2O | 1.23 (0.06) | −9.0 | 4.7 | |
| GHM7 | 1 | 800 | 24 | H2O-NaCl | 1.50 (0.31) | −9.0 | 4.7 | |
| GHM4 | 1 | 900 | 24 | H2O | 1.82 (0.11) | −7.1 | 4.7 | |
| GHM11 | 1 | 900 | 24 | H2O-NaCl | 2.17 (0.26) | −7.1 | 4.7 | |
| GM1* | 1 | 900 | 24 | H2O | 1.61 (0.09) | - | - | 0.64 ± 0.15 |
| GH2* | 1 | 900 | 24 | H2O | 1.42 (0.13) | - | - | 0.73 ± 0.22 |
| GHM13* | 1 | 950 | 24 | H2O | 1.96 (0.04) | −6.4 | 4.8 | |
| GHM15* | 1 | 950 | 24 | H2O-NaCl | 2.40 (0.12) | −6.4 | 4.8 | |
| GHM19 | 2 | 700 | 48 | H2O-NaCl | 0.60 (0.09) | −11.3 | 3.8 | |
| GHM20* | 2 | 800 | 24 | H2O | 0.96 (0.11) | −8.9 | 3.9 | |
| GHM9* | 2 | 900 | 24 | H2O | 1.85 (0.18) | −6.9 | 4.0 | 0.79 ± 0.04 |
| GHM10* | 2 | 900 | 24 | H2O-NaCl | 2.07 (0.41) | −6.9 | 4.0 | 0.45 ± 0.07 |
Notes: #ΔFMQ = logfO2run – logfO2FMQ (Fayalite+Magnetite+Quartz).
*Runs employing single crystals as starting materials. GM1 and GH2 denote runs with only magnetite or hematite (unbuffered conditions).Uncertainties (in parenthesis) given as 1 standard deviation of
n (10 < n < 50) EMP analyses of the glass.
Measured FeO contents in the quenched glasses from the H2O + melt system (Fig. 2a) range from 0.60 ± 0.21 wt. % at 1 GPa and 700 °C to 1.96 ± 0.04 wt. % at 1 GPa and 950 °C. In the H2O-NaCl + melt system (Fig. 2b), the FeO content increases from 0.75 ± 0.08 wt. % at 1 GPa and 700 °C to 2.40 ± 0.12 wt. % at 1 GPa and 950 °C. Within error, similar FeO contents were measured in glasses quenched at 2 GPa in both pure H2O and saline systems. Hence there is no significant pressure effect on iron solubility in agreement with previous low-pressure experiments in melt-free HCl-bearing fluids (Scholten et al., 2019
Scholten, L., Schmidt, C., Lecumberri-Sanchez, P., Newville, M., Lanzirotti, A., Sirbescu, M.L.C., Steele-MacInnis, M. (2019) Solubility and speciation of iron in hydrothermal fluids. Geochimica et Cosmochimica Acta 252, 126–143. https://doi.org/10.1016/j.gca.2019.03.001
). The two experimental runs performed with either magnetite or hematite in the presence of a hydrous melt phase displayed lower FeO contents in the quenched glass at 1 GPa and 900 °C compared to experiments in which both oxides were present, i.e. 1.61 ± 0.09 wt. % and 1.42 ± 0.13 wt. %, respectively (Fig. 2a). The FeO contents reported here compare well with the results from sediment melting experiments at similar conditions (e.g., Hermann and Spandler, 2008Hermann, J., Spandler, C.J. (2008) Sediment melts at sub-arc depths: An experimental study. Journal of Petrology 49, 717–740. https://doi.org/10.1093/petrology/egm073
) and from natural melt inclusions in hematite–magnetite-bearing mantle xenoliths (e.g., Blatter and Carmichael, 1998Blatter, D.L., Carmichael, I.S.E. (1998) Hornblende peridotite xenoliths from central Mexico reveal the highly oxidized nature of subarc upper mantle. Geology 26, 1035–1038. https://doi.org/10.1130/0091-7613(1998)026%3C1035:HPXFCM%3E2.3.CO;2
). A more detailed comparison is provided in the Supplementary Information.
Figure 2 Experimental FeO contents (wt. %) measured by EMPA in the quenched glasses, as a function of temperature for (a) the H2O + melt system and (b) the H2O-NaCl + melt system.
Mössbauer absorption spectra recorded on selected recovered glasses (i.e. with the highest FeO content) yielded Fe3+/Fetot ratios of 0.79 ± 0.04 in the H2O + haplogranite melt system (Fig. S-2a), while a lower Fe3+/Fetot ratio of 0.45 ± 0.07 was retrieved in the H2O-NaCl + melt system (Fig. S-2b), both at 2 GPa and 900 °C. Unbuffered experiments at 1 GPa and 900 °C showed Fe3+/Fetot ratios of 0.64 ± 0.15 and 0.73 ± 0.22 for the magnetite-only and hematite-only experiments, respectively (Fig. S-2c and S-2d). Additional details about the coordination environment of Fe3+ in the melt structure and the two unbuffered experimental runs are discussed in the Supplementary Information (Fig. S-2, S-3, and Table S-2).
top
Aqueous Fluids vs. Hydrous Silicate Melts as Carriers of Oxidised Iron
We show that significant amounts of iron, preferentially as Fe3+, can be mobilised and transported by hydrous silicate melts at sub-arc conditions. To compare the relative iron transport capacity of aqueous fluids and hydrous silicate melts, we further performed equilibrium thermodynamic calculations using the DEW model (Sverjensky et al., 2014
Sverjensky, D.A., Harrison, B., Azzolini, D. (2014) Water in the deep Earth: The dielectric constant and the solubilities of quartz and corundum to 60kb and 1200°C. Geochimica et Cosmochimica Acta 129, 125–145. https://doi.org/10.1016/j.gca.2013.12.019
) and computed the iron content (and speciation) in the aqueous phase at run conditions (see Supplementary Information).Thermodynamic modelling results indicate that hematite and magnetite are insoluble in H2O-only fluids at the investigated P, T, and fO2 conditions (Table S-3), with iron concentrations that are several orders of magnitude lower than those measured in the quenched glass (Fig. 3a). However, the stepwise addition of silica as Si(OH)4, following the haplogranite melt dissolution into the fluid, significantly increases the FeO content compared to the H2O-only system, due to the formation of a hydrous Fe-Si complex with stoichiometry Fe(H3SiO4)+ (Fig. 3a). Dissolved silicate species (Manning et al., 2010
Manning, C.E., Antignano, A., Lin, H.A. (2010) Premelting polymerization of crustal and mantle fluids, as indicated by the solubility of albite+paragonite+quartz in H2O at 1 GPa and 350-620°C. Earth and Planetary Science Letters 292, 325–336. https://doi.org/10.1016/j.epsl.2010.01.044
) thus provide additional ligands that enhance the mobilisation and transport of iron as previously reported for other ‘immobile’ metals, such as Zr, Ti, Nb (Audétat and Keppler, 2005Audétat, A., Keppler, H. (2005) Solubility of rutile in subduction zone fluids, as determined by experiments in the hydrothermal diamond anvil cell. Earth and Planetary Science Letters 232, 393–402. https://doi.org/10.1016/j.epsl.2005.01.028
; Manning et al., 2008Manning, C.E., Wilke, M., Schmidt, C., Cauzid, J. (2008) Rutile solubility in albite-H2O and Na2Si3O7-H2O at high temperatures and pressures by in-situ synchrotron radiation micro-XRF. Earth and Planetary Science Letters 272, 730–737. https://doi.org/10.1016/j.epsl.2008.06.004
; Wilke et al., 2012Wilke, M., Schmidt, C., Dubrail, J., Appel, K., Borchert, M., Kvashnina, K., Manning, C.E. (2012) Zircon solubility and zirconium complexation in H2O+Na2O+SiO2±Al2O3 fluids at high pressure and temperature. Earth and Planetary Science Letters 349–350, 15–25. https://doi.org/10.1016/j.epsl.2012.06.054
; Louvel et al., 2013Louvel, M., Sanchez-Valle, C., Malfait, W.J., Testemale, D., Hazemann, J.L. (2013) Zr complexation in high pressure fluids and silicate melts and implications for the mobilization of HFSE in subduction zones. Geochimica et Cosmochimica Acta 104, 281–299. https://doi.org/10.1016/j.gca.2012.11.001
) and most recently, for carbon in graphite-saturated systems (Tiraboschi et al., 2022Tiraboschi, C., Miozzi, F., Tumiati, S. (2022) Carbon-saturated COH fluids in the upper mantle: a review of high-pressure and high-temperature ex situ experiments. European Journal of Mineralogy 34, 59–75. https://doi.org/10.5194/ejm-34-59-2022
and references therein). In H2O-NaCl fluids, in contrast, the formation of FeCl2 complexes yields iron concentrations in the fluid in equilibrium with hematite–magnetite that are 4 orders of magnitude higher than in pure H2O (Fig. 3b and Table S-3). The addition of dissolved silica in the model further enhances the solubility of iron via the formation of Fe(H3SiO4)+ species that, together with Cl-complexes, lead to concentrations of Fe2+ dissolved in the fluid that are up to 5 times higher compared to the NaCl-free system.
Figure 3 Calculated hematite–magnetite solubility (DEW model, Sverjensky et al., 2014
Sverjensky, D.A., Harrison, B., Azzolini, D. (2014) Water in the deep Earth: The dielectric constant and the solubilities of quartz and corundum to 60kb and 1200°C. Geochimica et Cosmochimica Acta 129, 125–145. https://doi.org/10.1016/j.gca.2013.12.019
) in (a) aqueous and (b) saline fluids at 1 GPa, and comparison with experimental results in silicate melts. Dark grey diamonds: FeO content (mol/kg) in the H2O-only and H2O-NaCl system. Light grey diamonds: FeO (mol/kg) in Si(OH)4-bearing fluids (mol/kg, pink boxes). Circles (H2O-only) and triangles (H2O-NaCl) denote experimental FeO contents in hydrous silicate melts (mol/kg) from this study. Error bars are smaller than the symbol size. The black double-ended arrow shows the effect of oxidised iron. Coloured fields denote the dominance of the indicated solute species in the fluid.In both H2O and H2O-NaCl systems, the calculated FeO solute content in the fluid is 1 order of magnitude lower than in quenched glasses, hence indicating a higher iron transport capacity of melt compared to aqueous and saline fluids. Moreover, the Mössbauer data indicate that the higher FeO content in the quenched glass is mainly due to the presence of oxidised iron that dominates in the melt phase (Table 1 and Fig. 3). Taking into account the total FeO content obtained by EMP analyses (Table 1 and Table S-1) and the Mössbauer Fe3+/Fetot ratio, the estimated Fe2O3 content in the quenched glasses corresponds to 1.59 wt. % and 0.98 wt. % for the pure H2O and H2O-NaCl systems, respectively. The lower Fe2O3 content in the saline system can be partially attributed to the higher abundance of Fe2+, due to the higher solubility of hematite and magnetite in H2O-NaCl fluids (i.e. 0.118 mol/kg), i.e. two times the calculated solubility in the H2O-only system (0.057 mol/kg).
top
Mobilisation and Transfer of Oxidised Iron at Sub-Arc Conditions
Our experimental results show that hematite and magnetite can be effectively dissolved by hydrous silicate melts and provide the first constraints of the amount of oxidised iron transferred by hydrous melts at sub-arc conditions. As the Fe3+/Fetot ratio in silicate melts increases upon increasing temperature and oxygen fugacity (Wilke et al., 2002
Wilke, M., Behrens, H., Burkhard, D.J.M., Rossano, S. (2002) The oxidation state of iron in silicic melt at 500 MPa water pressure. Chemical Geology 189, 55–67. https://doi.org/10.1016/S0009-2541(02)00042-6
; Borisov and McCammon, 2010Borisov, A., McCammon, C. (2010) The effect of silica on ferric/ferrous ratio in silicate melts: An experimental study using Mössbauer spectroscopy. American Mineralogist 95, 545–555. https://doi.org/10.2138/am.2010.3217
; Mysen and Richet, 2019Mysen, B., Richet, P. (2019) Chapter 11 - Structure of Iron Silicate Glasses and Melts. In: Mysen, B., Richet, P. (Eds.) Silicate Glasses and Melts. Second Edition, Elsevier, Amsterdam, Oxford, Cambridge, 403–430. https://doi.org/10.1016/B978-0-444-63708-6.00011-9
), our results at 2 GPa and 900 °C (0.98–1.59 wt. % Fe2O3) likely represent the upper limit for Fe3+ transferred by hydrous melts as the temperature is higher than typical conditions for most hot subduction regimes (Fig. 1). Applying correlations between the Fe3+/Fe2+ ratio and the oxygen fugacity (see details in the Supplementary Information, Fig. S-4), we estimate the Fe2O3 content in hydrous silicate melts at sub-arc conditions in hot subduction slabs, i.e. 2 GPa and 700 °C (Fig. 1), to be 0.15–0.23 wt. % and 0.05–0.08 wt. % for pure H2O and saline systems, respectively. The amount of oxidised iron mobilised by the dissolution of hematite–magnetite assemblages in hydrous melts ranges between 0.15 to 1.59 wt. % (0.05 to 0.98 wt. % in saline systems) Fe2O3 for the range of hot subduction slab geotherms. The dissolution of ‘fluid-insoluble’ iron oxides in hydrous slab-melts may thus have a significant contribution to the iron budget and redox capacity of the slab flux and should be considered when estimating the redox state of hot subduction zones.Despite the simplicity of the system investigated here, our results have direct implications for subduction environments where melting of hematite and magnetite-bearing metasediments occur (Ague et al., 2022
Ague, J.J., Tassara, S., Holycross, M.E., Li, J.-L., Cottrell, E., Schwarzenbach, E.M., Fassoulas, C., John, T. (2022) Slab-derived devolatilization fluids oxidized by subducted metasedimentary rocks. Nature Geoscience 15, 320–326. https://doi.org/10.1038/s41561-022-00904-7
) or where hydrous silicate melts produced by metasediment slices interact with serpentine in subduction mélanges (Marschall and Schumacher, 2012Marschall, H.R., Schumacher, J.C. (2012) Arc magmas sourced from mélange diapirs in subduction zones. Nature Geoscience 5, 862–867. https://doi.org/10.1038/ngeo1634
). Hydrous silicate melts are thus efficient agents for the mobilisation and transfer of oxidised iron from the slab to the mantle wedge, ultimately contributing to the oxidation of the arc magma source.top
Acknowledgements
This work was supported by the German Research Foundation (DFG) through grants TI 1078/2-1 and SA 2585/4-1. C.T. acknowledges a mobility grant from the Italian Society of Mineralogy and Petrography (SIMP). S. Flemetakis and Z. Liu are thanked for their assistance in the laboratory, M. Trogisch and B. Schmitte for sample preparation, and D. Sverjensky and S. Tumiati for help with the thermodynamic modelling. Katy Evans and Antoine Bénard are acknowledged for helpful reviews and Horst Marschall for editorial handling.
Editor: Horst R. Marschall
top
References
Ague, J.J., Tassara, S., Holycross, M.E., Li, J.-L., Cottrell, E., Schwarzenbach, E.M., Fassoulas, C., John, T. (2022) Slab-derived devolatilization fluids oxidized by subducted metasedimentary rocks. Nature Geoscience 15, 320–326. https://doi.org/10.1038/s41561-022-00904-7
 Show in context
Show in context Iron oxides such as hematite, Fe2O3, and magnetite, Fe3O4, are by-products of serpentinisation (e.g., Carbonin et al., 2015; Debret et al., 2015), and they have been identified in seafloor metasediments (e.g., Ague et al., 2022).
View in article
Despite the simplicity of the system investigated here, our results have direct implications for subduction environments where melting of hematite and magnetite-bearing metasediments occur (Ague et al., 2022) or where hydrous silicate melts produced by metasediment slices interact with serpentine in subduction mélanges (Marschall and Schumacher, 2012).
View in article
Audétat, A., Keppler, H. (2005) Solubility of rutile in subduction zone fluids, as determined by experiments in the hydrothermal diamond anvil cell. Earth and Planetary Science Letters 232, 393–402. https://doi.org/10.1016/j.epsl.2005.01.028
 Show in context
Show in context Dissolved silicate species (Manning et al., 2010) thus provide additional ligands that enhance the mobilisation and transport of iron as previously reported for other ‘immobile’ metals, such as Zr, Ti, Nb (Audétat and Keppler, 2005; Manning et al., 2008; Wilke et al., 2012; Louvel et al., 2013) and most recently, for carbon in graphite-saturated systems (Tiraboschi et al., 2022 and references therein).
View in article
Bell, A.S., Webster, J.D. (2015) Dissolved Cl, oxygen fugacity, and their effects on Fe behavior in a hydrous rhyodacitic melt. American Mineralogist 100, 1595–1599. https://doi.org/10.2138/am-2015-5197
 Show in context
Show in context To date, most experimental studies on the solubility of hematite and/or magnetite in aqueous fluids (Chou and Eugster, 1977; Boctor et al., 1980; Frantz et al., 1981; Scholten et al., 2019) and melts (Simon et al., 2004; Bell and Webster, 2015), have been conducted at moderate pressures, less than 0.4 GPa, that are not relevant for sub-arc settings.
View in article
Bénard, A., Klimm, K., Woodland, A.B., Arculus, R.J., Wilke, M., Botcharnikov, R.E., Shimizu, N., Nebel, O., Rivard, C., Ionov, D.A. (2018) Oxidising agents in sub-arc mantle melts link slab devolatilisation and arc magmas. Nature Communications 9, 1–10. https://doi.org/10.1038/s41467-018-05804-2
 Show in context
Show in context The oxidised signature is thought to arise from the influx of oxidising slab-derived fluids or melts into the mantle wedge (e.g., Burgisser and Scaillet, 2007; Debret and Sverjensky, 2017; Bénard et al., 2018; Walters et al., 2020; Evans and Frost, 2021), although the actual oxidising agent and the transport mechanism remain a topic of discussion (e.g., Piccoli et al., 2019; Tollan and Hermann, 2019).
View in article
Alternatively, silica-bearing melt-like fluids have been suggested as an efficient agent for the transfer of oxidised iron (Parkinson and Arculus, 1999; Hermann et al., 2006; Kelley and Cottrell, 2009; Bénard et al., 2018) owing to its low solubility in aqueous phases (Wykes et al., 2008).
View in article
Blatter, D.L., Carmichael, I.S.E. (1998) Hornblende peridotite xenoliths from central Mexico reveal the highly oxidized nature of subarc upper mantle. Geology 26, 1035–1038. https://doi.org/10.1130/0091-7613(1998)026%3C1035:HPXFCM%3E2.3.CO;2
 Show in context
Show in context The FeO contents reported here compare well with the results from sediment melting experiments at similar conditions (e.g., Hermann and Spandler, 2008) and from natural melt inclusions in hematite–magnetite-bearing mantle xenoliths (e.g., Blatter and Carmichael, 1998).
View in article
Boctor, N.Z., Popp, R.K., Frantz, J.D. (1980) Mineral-solution equilibria—IV. Solubilities and the thermodynamic properties of FeCl20 in the system Fe2O3-H2-H2O-HCl. Geochimica et Cosmochimica Acta 44, 1509–1518. https://doi.org/10.1016/0016-7037(80)90115-5
 Show in context
Show in context To date, most experimental studies on the solubility of hematite and/or magnetite in aqueous fluids (Chou and Eugster, 1977; Boctor et al., 1980; Frantz et al., 1981; Scholten et al., 2019) and melts (Simon et al., 2004; Bell and Webster, 2015), have been conducted at moderate pressures, less than 0.4 GPa, that are not relevant for sub-arc settings.
View in article
Borisov, A., McCammon, C. (2010) The effect of silica on ferric/ferrous ratio in silicate melts: An experimental study using Mössbauer spectroscopy. American Mineralogist 95, 545–555. https://doi.org/10.2138/am.2010.3217
 Show in context
Show in context As the Fe3+/Fetot ratio in silicate melts increases upon increasing temperature and oxygen fugacity (Wilke et al., 2002; Borisov and McCammon, 2010; Mysen and Richet, 2019), our results at 2 GPa and 900 °C (0.98–1.59 wt. % Fe2O3) likely represent the upper limit for Fe3+ transferred by hydrous melts as the temperature is higher than typical conditions for most hot subduction regimes (Fig. 1).
View in article
Brounce, M.N., Kelley, K.A., Cottrell, E. (2014) Variations in Fe3+/∑Fe of Mariana Arc Basalts and Mantle Wedge fO2. Journal of Petrology 55, 2514–2536. https://doi.org/10.1093/petrology/egu065
 Show in context
Show in context Arc magmas are characterised by a higher oxidation degree than magmas from other tectonic settings, including mid-oceanic ridge and ocean arc basalts (Carmichael, 1991; Kelley and Cottrell, 2009; Brounce et al., 2014).
View in article
Bureau, H., Keppler, H. (1999) Complete miscibility between silicate melts and hydrous fluids in the upper mantle: Experimental evidence and geochemical implications. Earth and Planetary Science Letters 165, 187–196. https://doi.org/10.1016/S0012-821X(98)00266-0
 Show in context
Show in context The dotted line denotes the critical curve in the haplogranite–H2O system (Bureau and Keppler, 1999).
View in article
Burgisser, A., Scaillet, B. (2007) Redox evolution of a degassing magma rising to the surface. Nature 445, 194–197. https://doi.org/10.1038/nature05509
 Show in context
Show in context The oxidised signature is thought to arise from the influx of oxidising slab-derived fluids or melts into the mantle wedge (e.g., Burgisser and Scaillet, 2007; Debret and Sverjensky, 2017; Bénard et al., 2018; Walters et al., 2020; Evans and Frost, 2021), although the actual oxidising agent and the transport mechanism remain a topic of discussion (e.g., Piccoli et al., 2019; Tollan and Hermann, 2019).
View in article
Carbonin, S., Martin, S., Tumiati, S., Rossetti, P. (2015) Magnetite from the Cogne serpentinites (Piemonte ophiolite nappe, Italy). Insights into seafloor fluid–rock interaction. European Journal of Mineralogy 27, 31–50. https://doi.org/10.1127/ejm/2014/0026-2410
 Show in context
Show in context Iron oxides such as hematite, Fe2O3, and magnetite, Fe3O4, are by-products of serpentinisation (e.g., Carbonin et al., 2015; Debret et al., 2015), and they have been identified in seafloor metasediments (e.g., Ague et al., 2022).
View in article
Carmichael, I.S.E. (1991) The redox states of basic and silicic magmas: a reflection of their source regions? Contributions to Mineralogy and Petrology 106, 129–141. https://doi.org/10.1007/BF00306429
 Show in context
Show in context Arc magmas are characterised by a higher oxidation degree than magmas from other tectonic settings, including mid-oceanic ridge and ocean arc basalts (Carmichael, 1991; Kelley and Cottrell, 2009; Brounce et al., 2014).
View in article
Chou, I.M., Eugster, H.P. (1977) Solubility of magnetite in supercritical chloride solutions. American Journal of Science 277, 1296–1314. https://doi.org/10.2475/ajs.277.10.1296
 Show in context
Show in context To date, most experimental studies on the solubility of hematite and/or magnetite in aqueous fluids (Chou and Eugster, 1977; Boctor et al., 1980; Frantz et al., 1981; Scholten et al., 2019) and melts (Simon et al., 2004; Bell and Webster, 2015), have been conducted at moderate pressures, less than 0.4 GPa, that are not relevant for sub-arc settings.
View in article
Debret, B., Sverjensky, D.A. (2017) Highly oxidising fluids generated during serpentinite breakdown in subduction zones. Scientific Reports 7, 1–6. https://doi.org/10.1038/s41598-017-09626-y
 Show in context
Show in context The oxidised signature is thought to arise from the influx of oxidising slab-derived fluids or melts into the mantle wedge (e.g., Burgisser and Scaillet, 2007; Debret and Sverjensky, 2017; Bénard et al., 2018; Walters et al., 2020; Evans and Frost, 2021), although the actual oxidising agent and the transport mechanism remain a topic of discussion (e.g., Piccoli et al., 2019; Tollan and Hermann, 2019).
View in article
Debret, B., Bolfan-Casanova, N., Padron-Navarta, J.A., Martin-Hernandez, F., Andreani, M., Garrido, C.J., Sánchez-Vizcaíno V.L., Gómez-Pugnaire M.T., Mu˜noz, M., Trcera, N. (2015) Redox state of iron during high-pressure serpentinite dehydration. Contributions to Mineralogy and Petrology 169, 1–18. https://doi.org/10.1007/s00410-015-1130-y
 Show in context
Show in context Iron oxides such as hematite, Fe2O3, and magnetite, Fe3O4, are by-products of serpentinisation (e.g., Carbonin et al., 2015; Debret et al., 2015), and they have been identified in seafloor metasediments (e.g., Ague et al., 2022).
View in article
Evans, K.A. (2012) The redox budget of subduction zones. Earth-Science Reviews 113, 11–32. https://doi.org/10.1016/j.earscirev.2012.03.003
 Show in context
Show in context Iron, as the most abundant redox-sensitive element in the upper mantle, may play a pivotal role in these processes, and constraints on its mobility and speciation in slab-derived phases are thus critical to elucidating the range of redox state conditions of subduction zones (Frost and McCammon, 2008; Kelley and Cottrell, 2009; Evans, 2012).
View in article
This value implies Fe3+ fluxes of 109 mol/year, which are three to four orders of magnitude lower than the estimated redox budget flux (55 ± 13 × 1012 mol/year; Evans, 2012).
View in article
Evans, K.A., Frost, B.R. (2021) Deserpentinization in subduction zones as a source of oxidation in arcs: A reality check. Journal of Petrology 62, 1–32. https://doi.org/10.1093/petrology/egab016
 Show in context
Show in context The oxidised signature is thought to arise from the influx of oxidising slab-derived fluids or melts into the mantle wedge (e.g., Burgisser and Scaillet, 2007; Debret and Sverjensky, 2017; Bénard et al., 2018; Walters et al., 2020; Evans and Frost, 2021), although the actual oxidising agent and the transport mechanism remain a topic of discussion (e.g., Piccoli et al., 2019; Tollan and Hermann, 2019).
View in article
Frantz, J.D., Popp, R.K., Boctor, N.Z. (1981) Mineral-solution equilibria-V. Solubilities of rock-forming minerals in supercritical fluids. Geochimica et Cosmochimica Acta 45, 69–77. https://doi.org/10.1016/0016-7037(81)90264-7
 Show in context
Show in context To date, most experimental studies on the solubility of hematite and/or magnetite in aqueous fluids (Chou and Eugster, 1977; Boctor et al., 1980; Frantz et al., 1981; Scholten et al., 2019) and melts (Simon et al., 2004; Bell and Webster, 2015), have been conducted at moderate pressures, less than 0.4 GPa, that are not relevant for sub-arc settings.
View in article
Frost, D.J., McCammon, C.A. (2008) The Redox State of Earth’s Mantle. Annual Review of Earth and Planetary Sciences 36, 389–420. https://doi.org/10.1146/annurev.earth.36.031207.124322
 Show in context
Show in context Iron, as the most abundant redox-sensitive element in the upper mantle, may play a pivotal role in these processes, and constraints on its mobility and speciation in slab-derived phases are thus critical to elucidating the range of redox state conditions of subduction zones (Frost and McCammon, 2008; Kelley and Cottrell, 2009; Evans, 2012).
View in article
Hermann, J., Spandler, C.J. (2008) Sediment melts at sub-arc depths: An experimental study. Journal of Petrology 49, 717–740. https://doi.org/10.1093/petrology/egm073
 Show in context
Show in context The FeO contents reported here compare well with the results from sediment melting experiments at similar conditions (e.g., Hermann and Spandler, 2008) and from natural melt inclusions in hematite–magnetite-bearing mantle xenoliths (e.g., Blatter and Carmichael, 1998).
View in article
Hermann, J., Spandler, C., Hack, A., Korsakov, A.V. (2006) Aqueous fluids and hydrous melts in high-pressure and ultra-high pressure rocks: Implications for element transfer in subduction zones. Lithos 92, 399–417. https://doi.org/10.1016/j.lithos.2006.03.055
 Show in context
Show in context Alternatively, silica-bearing melt-like fluids have been suggested as an efficient agent for the transfer of oxidised iron (Parkinson and Arculus, 1999; Hermann et al., 2006; Kelley and Cottrell, 2009; Bénard et al., 2018) owing to its low solubility in aqueous phases (Wykes et al., 2008).
View in article
Kelley, K.A., Cottrell, E. (2009) Water and the oxidation state of subduction zone magmas. Science 325, 605–607. https://doi.org/10.1126/science.1174156
 Show in context
Show in context Arc magmas are characterised by a higher oxidation degree than magmas from other tectonic settings, including mid-oceanic ridge and ocean arc basalts (Carmichael, 1991; Kelley and Cottrell, 2009; Brounce et al., 2014).
View in article
Iron, as the most abundant redox-sensitive element in the upper mantle, may play a pivotal role in these processes, and constraints on its mobility and speciation in slab-derived phases are thus critical to elucidating the range of redox state conditions of subduction zones (Frost and McCammon, 2008; Kelley and Cottrell, 2009; Evans, 2012).
View in article
Alternatively, silica-bearing melt-like fluids have been suggested as an efficient agent for the transfer of oxidised iron (Parkinson and Arculus, 1999; Hermann et al., 2006; Kelley and Cottrell, 2009; Bénard et al., 2018) owing to its low solubility in aqueous phases (Wykes et al., 2008).
View in article
The dissolution of iron oxides in slab-derived silicate melts thus provides a viable mechanism for the transfer of oxidised iron from the slab to the mantle wedge and to explain the oxidised nature of arc magmas (Kelley and Cottrell, 2009).
View in article
Louvel, M., Sanchez-Valle, C., Malfait, W.J., Testemale, D., Hazemann, J.L. (2013) Zr complexation in high pressure fluids and silicate melts and implications for the mobilization of HFSE in subduction zones. Geochimica et Cosmochimica Acta 104, 281–299. https://doi.org/10.1016/j.gca.2012.11.001
 Show in context
Show in context Dissolved silicate species (Manning et al., 2010) thus provide additional ligands that enhance the mobilisation and transport of iron as previously reported for other ‘immobile’ metals, such as Zr, Ti, Nb (Audétat and Keppler, 2005; Manning et al., 2008; Wilke et al., 2012; Louvel et al., 2013) and most recently, for carbon in graphite-saturated systems (Tiraboschi et al., 2022 and references therein).
View in article
Manning, C.E., Wilke, M., Schmidt, C., Cauzid, J. (2008) Rutile solubility in albite-H2O and Na2Si3O7-H2O at high temperatures and pressures by in-situ synchrotron radiation micro-XRF. Earth and Planetary Science Letters 272, 730–737. https://doi.org/10.1016/j.epsl.2008.06.004
 Show in context
Show in context Dissolved silicate species (Manning et al., 2010) thus provide additional ligands that enhance the mobilisation and transport of iron as previously reported for other ‘immobile’ metals, such as Zr, Ti, Nb (Audétat and Keppler, 2005; Manning et al., 2008; Wilke et al., 2012; Louvel et al., 2013) and most recently, for carbon in graphite-saturated systems (Tiraboschi et al., 2022 and references therein).
View in article
Manning, C.E., Antignano, A., Lin, H.A. (2010) Premelting polymerization of crustal and mantle fluids, as indicated by the solubility of albite+paragonite+quartz in H2O at 1 GPa and 350-620°C. Earth and Planetary Science Letters 292, 325–336. https://doi.org/10.1016/j.epsl.2010.01.044
 Show in context
Show in context Dissolved silicate species (Manning et al., 2010) thus provide additional ligands that enhance the mobilisation and transport of iron as previously reported for other ‘immobile’ metals, such as Zr, Ti, Nb (Audétat and Keppler, 2005; Manning et al., 2008; Wilke et al., 2012; Louvel et al., 2013) and most recently, for carbon in graphite-saturated systems (Tiraboschi et al., 2022 and references therein).
View in article
Marschall, H.R., Schumacher, J.C. (2012) Arc magmas sourced from mélange diapirs in subduction zones. Nature Geoscience 5, 862–867. https://doi.org/10.1038/ngeo1634
 Show in context
Show in context Despite the simplicity of the system investigated here, our results have direct implications for subduction environments where melting of hematite and magnetite-bearing metasediments occur (Ague et al., 2022) or where hydrous silicate melts produced by metasediment slices interact with serpentine in subduction mélanges (Marschall and Schumacher, 2012).
View in article
Mysen, B., Richet, P. (2019) Chapter 11 - Structure of Iron Silicate Glasses and Melts. In: Mysen, B., Richet, P. (Eds.) Silicate Glasses and Melts. Second Edition, Elsevier, Amsterdam, Oxford, Cambridge, 403–430. https://doi.org/10.1016/B978-0-444-63708-6.00011-9
 Show in context
Show in context As the Fe3+/Fetot ratio in silicate melts increases upon increasing temperature and oxygen fugacity (Wilke et al., 2002; Borisov and McCammon, 2010; Mysen and Richet, 2019), our results at 2 GPa and 900 °C (0.98–1.59 wt. % Fe2O3) likely represent the upper limit for Fe3+ transferred by hydrous melts as the temperature is higher than typical conditions for most hot subduction regimes (Fig. 1).
View in article
Parkinson, I.J., Arculus, R.J. (1999) The redox state of subduction zones: Insights from arc-peridotites. Chemical Geology 160, 409–423. https://doi.org/10.1016/S0009-2541(99)00110-2
 Show in context
Show in context Alternatively, silica-bearing melt-like fluids have been suggested as an efficient agent for the transfer of oxidised iron (Parkinson and Arculus, 1999; Hermann et al., 2006; Kelley and Cottrell, 2009; Bénard et al., 2018) owing to its low solubility in aqueous phases (Wykes et al., 2008).
View in article
Piccoli, F., Hermann, J., Pettke, T., Connolly, J.A.D., Kempf, E.D., Vieira Duarte, J.F. (2019) Subducting serpentinites release reduced, not oxidized, aqueous fluids. Scientific Reports 9, 1–7. https://doi.org/10.1038/s41598-019-55944-8
 Show in context
Show in context The oxidised signature is thought to arise from the influx of oxidising slab-derived fluids or melts into the mantle wedge (e.g., Burgisser and Scaillet, 2007; Debret and Sverjensky, 2017; Bénard et al., 2018; Walters et al., 2020; Evans and Frost, 2021), although the actual oxidising agent and the transport mechanism remain a topic of discussion (e.g., Piccoli et al., 2019; Tollan and Hermann, 2019).
View in article
Scholten, L., Schmidt, C., Lecumberri-Sanchez, P., Newville, M., Lanzirotti, A., Sirbescu, M.L.C., Steele-MacInnis, M. (2019) Solubility and speciation of iron in hydrothermal fluids. Geochimica et Cosmochimica Acta 252, 126–143. https://doi.org/10.1016/j.gca.2019.03.001
 Show in context
Show in context To date, most experimental studies on the solubility of hematite and/or magnetite in aqueous fluids (Chou and Eugster, 1977; Boctor et al., 1980; Frantz et al., 1981; Scholten et al., 2019) and melts (Simon et al., 2004; Bell and Webster, 2015), have been conducted at moderate pressures, less than 0.4 GPa, that are not relevant for sub-arc settings.
View in article
Hence there is no significant pressure effect on iron solubility in agreement with previous low-pressure experiments in melt-free HCl-bearing fluids (Scholten et al., 2019).
View in article
Simon, A.C., Pettke, T., Candela, P.A., Piccoli, P.M., Heinrich, C.A. (2004) Magnetite solubility and iron transport in magmatic-hydrothermal environments. Geochimica et Cosmochimica Acta 68, 4905–4914. https://doi.org/10.1016/j.gca.2004.05.033
 Show in context
Show in context To date, most experimental studies on the solubility of hematite and/or magnetite in aqueous fluids (Chou and Eugster, 1977; Boctor et al., 1980; Frantz et al., 1981; Scholten et al., 2019) and melts (Simon et al., 2004; Bell and Webster, 2015), have been conducted at moderate pressures, less than 0.4 GPa, that are not relevant for sub-arc settings.
View in article
Sverjensky, D.A., Harrison, B., Azzolini, D. (2014) Water in the deep Earth: The dielectric constant and the solubilities of quartz and corundum to 60kb and 1200°C. Geochimica et Cosmochimica Acta 129, 125–145. https://doi.org/10.1016/j.gca.2013.12.019
 Show in context
Show in context To compare the relative iron transport capacity of aqueous fluids and hydrous silicate melts, we further performed equilibrium thermodynamic calculations using the DEW model (Sverjensky et al., 2014) and computed the iron content (and speciation) in the aqueous phase at run conditions (see Supplementary Information).
View in article
Calculated hematite–magnetite solubility (DEW model, Sverjensky et al., 2014) in (a) aqueous and (b) saline fluids at 1 GPa, and comparison with experimental results in silicate melts.
View in article
Syracuse, E.M., van Keken, P.E., Abers, G.A. (2010) The global range of subduction zone thermal models. Physics of the Earth and Planetary Interiors 183, 73–90. https://doi.org/10.1016/j.pepi.2010.02.004
 Show in context
Show in context Solid lines represent hot subduction geothermal paths (Syracuse et al., 2010).
View in article
Tiraboschi, C., Miozzi, F., Tumiati, S. (2022) Carbon-saturated COH fluids in the upper mantle: a review of high-pressure and high-temperature ex situ experiments. European Journal of Mineralogy 34, 59–75. https://doi.org/10.5194/ejm-34-59-2022
 Show in context
Show in context Dissolved silicate species (Manning et al., 2010) thus provide additional ligands that enhance the mobilisation and transport of iron as previously reported for other ‘immobile’ metals, such as Zr, Ti, Nb (Audétat and Keppler, 2005; Manning et al., 2008; Wilke et al., 2012; Louvel et al., 2013) and most recently, for carbon in graphite-saturated systems (Tiraboschi et al., 2022 and references therein).
View in article
Tollan, P., Hermann, J. (2019) Arc magmas oxidized by water dissociation and hydrogen incorporation in orthopyroxene. Nature Geoscience 12, 667–671. https://doi.org/10.1038/s41561-019-0411-x
 Show in context
Show in context The oxidised signature is thought to arise from the influx of oxidising slab-derived fluids or melts into the mantle wedge (e.g., Burgisser and Scaillet, 2007; Debret and Sverjensky, 2017; Bénard et al., 2018; Walters et al., 2020; Evans and Frost, 2021), although the actual oxidising agent and the transport mechanism remain a topic of discussion (e.g., Piccoli et al., 2019; Tollan and Hermann, 2019).
View in article
Walters, J.B., Cruz-Uribe, A.M., Marschall, H.R. (2020) Sulfur loss from subducted altered oceanic crust and implications for mantle oxidation. Geochemical Perspectives Letters 13, 36–41. https://doi.org/10.7185/geochemlet.2011
 Show in context
Show in context The oxidised signature is thought to arise from the influx of oxidising slab-derived fluids or melts into the mantle wedge (e.g., Burgisser and Scaillet, 2007; Debret and Sverjensky, 2017; Bénard et al., 2018; Walters et al., 2020; Evans and Frost, 2021), although the actual oxidising agent and the transport mechanism remain a topic of discussion (e.g., Piccoli et al., 2019; Tollan and Hermann, 2019).
View in article
Wilke, M., Behrens, H., Burkhard, D.J.M., Rossano, S. (2002) The oxidation state of iron in silicic melt at 500 MPa water pressure. Chemical Geology 189, 55–67. https://doi.org/10.1016/S0009-2541(02)00042-6
 Show in context
Show in context As the Fe3+/Fetot ratio in silicate melts increases upon increasing temperature and oxygen fugacity (Wilke et al., 2002; Borisov and McCammon, 2010; Mysen and Richet, 2019), our results at 2 GPa and 900 °C (0.98–1.59 wt. % Fe2O3) likely represent the upper limit for Fe3+ transferred by hydrous melts as the temperature is higher than typical conditions for most hot subduction regimes (Fig. 1).
View in article
Wilke, M., Schmidt, C., Dubrail, J., Appel, K., Borchert, M., Kvashnina, K., Manning, C.E. (2012) Zircon solubility and zirconium complexation in H2O+Na2O+SiO2±Al2O3 fluids at high pressure and temperature. Earth and Planetary Science Letters 349–350, 15–25. https://doi.org/10.1016/j.epsl.2012.06.054
 Show in context
Show in context Dissolved silicate species (Manning et al., 2010) thus provide additional ligands that enhance the mobilisation and transport of iron as previously reported for other ‘immobile’ metals, such as Zr, Ti, Nb (Audétat and Keppler, 2005; Manning et al., 2008; Wilke et al., 2012; Louvel et al., 2013) and most recently, for carbon in graphite-saturated systems (Tiraboschi et al., 2022 and references therein).
View in article
Wykes, J.L., Newton, R.C., Manning, C.E. (2008) Solubility of andradite, Ca3Fe2Si3O12, in a 10 mol% NaCl solution at 800 °C and 10 kbar: Implications for the metasomatic origin of grandite garnet in calc-silicate granulites. American Mineralogist 93, 886–892. https://doi.org/10.2138/am.2008.2736
 Show in context
Show in context A single study of hematite solubility in H2O-NaCl fluid at 1 GPa and 800 °C by Wykes et al. (2008) reported dissolved iron contents below 0.01 mol/kg.
View in article
Alternatively, silica-bearing melt-like fluids have been suggested as an efficient agent for the transfer of oxidised iron (Parkinson and Arculus, 1999; Hermann et al., 2006; Kelley and Cottrell, 2009; Bénard et al., 2018) owing to its low solubility in aqueous phases (Wykes et al., 2008).
View in article
top
Supplementary Information
The Supplementary Information includes:
- Materials and Methods
- Calculation of Fe3+ Transferred by Hydrous Melts in Subduction Zones
- Tables S-1 to S-3
- Figures S-1 to S-4
- Supplementary Information References
Download the Supplementary Information (PDF)
Figures
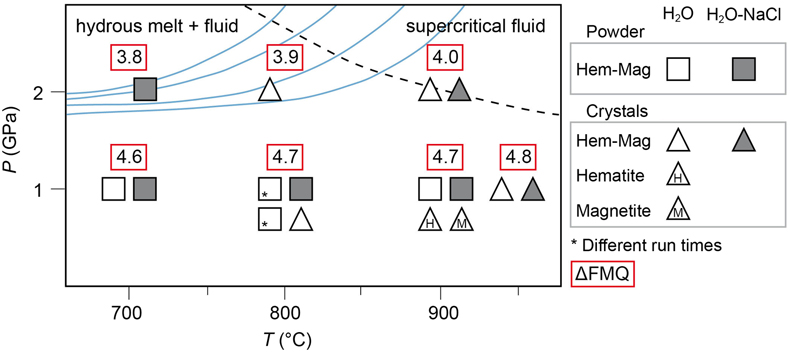
Figure 1 Pressure and temperature conditions of experimental runs employing powders (squares) or single crystals (triangles) as starting materials. The oxygen fugacity is reported relative to the Fayalite-Magnetite-Quartz (FMQ) buffer. The dotted line denotes the critical curve in the haplogranite–H2O system (Bureau and Keppler, 1999
Bureau, H., Keppler, H. (1999) Complete miscibility between silicate melts and hydrous fluids in the upper mantle: Experimental evidence and geochemical implications. Earth and Planetary Science Letters 165, 187–196. https://doi.org/10.1016/S0012-821X(98)00266-0
). Solid lines represent hot subduction geothermal paths (Syracuse et al., 2010Syracuse, E.M., van Keken, P.E., Abers, G.A. (2010) The global range of subduction zone thermal models. Physics of the Earth and Planetary Interiors 183, 73–90. https://doi.org/10.1016/j.pepi.2010.02.004
).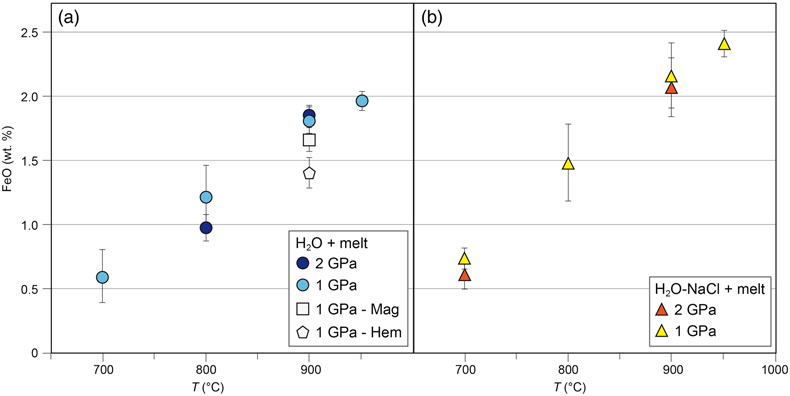
Figure 2 Experimental FeO contents (wt. %) measured by EMPA in the quenched glasses, as a function of temperature for (a) the H2O + melt system and (b) the H2O-NaCl + melt system.
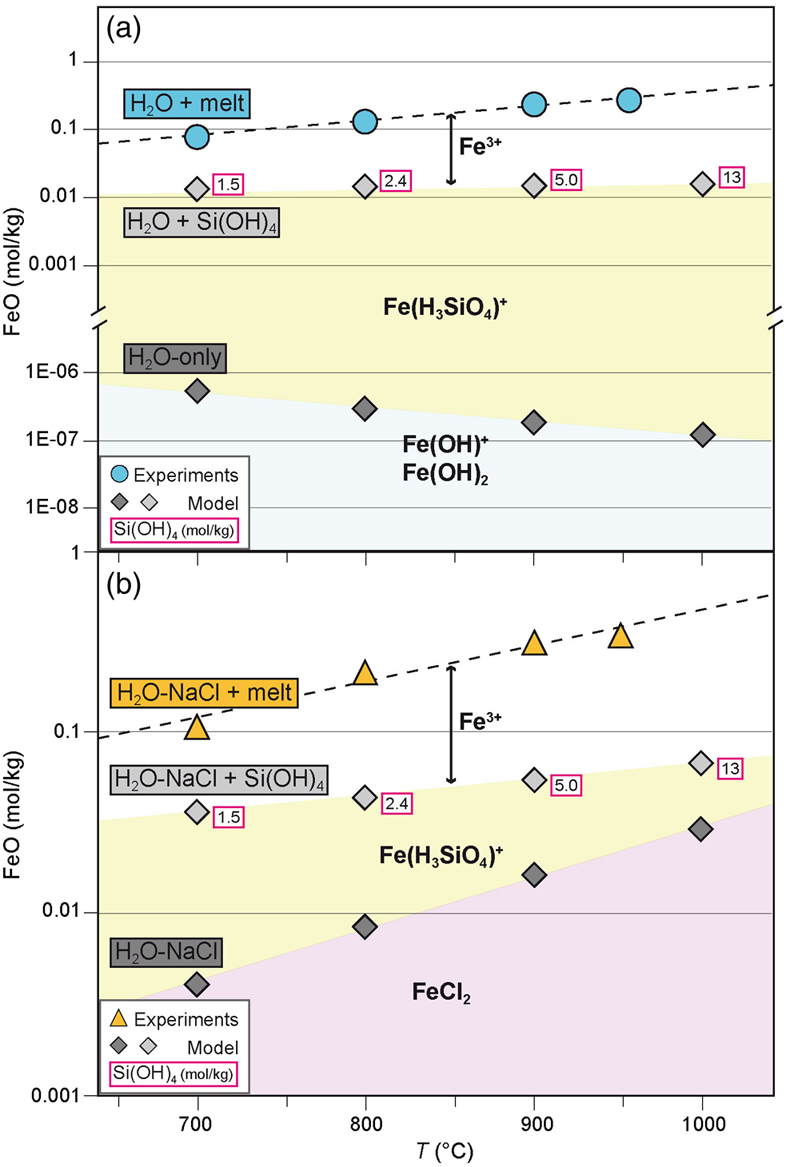
Figure 3 Calculated hematite–magnetite solubility (DEW model, Sverjensky et al., 2014
Sverjensky, D.A., Harrison, B., Azzolini, D. (2014) Water in the deep Earth: The dielectric constant and the solubilities of quartz and corundum to 60kb and 1200°C. Geochimica et Cosmochimica Acta 129, 125–145. https://doi.org/10.1016/j.gca.2013.12.019
) in (a) aqueous and (b) saline fluids at 1 GPa, and comparison with experimental results in silicate melts. Dark grey diamonds: FeO content (mol/kg) in the H2O-only and H2O-NaCl system. Light grey diamonds: FeO (mol/kg) in Si(OH)4-bearing fluids (mol/kg, pink boxes). Circles (H2O-only) and triangles (H2O-NaCl) denote experimental FeO contents in hydrous silicate melts (mol/kg) from this study. Error bars are smaller than the symbol size. The black double-ended arrow shows the effect of oxidised iron. Coloured fields denote the dominance of the indicated solute species in the fluid.