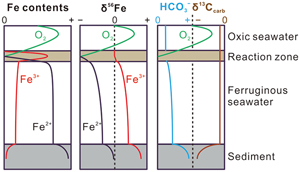
 | Isotope evidence for the coupled iron and carbon cycles 1.4 billion years ago Abstract: Although ferruginous waters were widespread in the deep ocean until 0.58 Ga, massive Fe deposits were rare during the interval from 1.8−0.8 Ga. Here, we report the contents and isotopic compositions of Fe and C from the ∼1.4 Ga Xiamaling (XML) siderite deposits, North China. A genetic model is introduced to interpret the dynamic and coupled Fe and C cycles in Mesoproterozoic oceans and sediments. Although the positive Eu anomalies of XML siderites are relatively low in the XML deposits, an enhanced input of hydrothermal Fe2+ is recognised from the isotopic compositions of Fe and C. Hydrothermal fluid is suggested as a key controlling factor during the XML siderite deposition. |
 | Early precipitated micropyrite in microbialites: A time capsule of microbial sulfur cycling Abstract: Microbialites are organosedimentary rocks that have occurred throughout the Earth’s history. The relationships between diverse microbial metabolic activities and isotopic signatures in biominerals forming within these microbialites are key to understanding modern biogeochemical cycles, but also for accurate interpretation of the geologic record. Here, we performed detailed mineralogical investigations coupled with NanoSIMS (Nanoscale Secondary Ion Mass Spectrometry) analyses of pyrite S isotopes in mineralising microbial mats from two different environments, a hypersaline lagoon (Cayo Coco, Cuba) and a volcanic alkaline crater lake (Atexcac, Mexico). Both microbialite samples contain two distinct pyrite morphologies: framboids and euhedral micropyrites, which display distinct ranges of δ34S values1. Considering the sulfate-sulfur isotopic compositions associated with both environments, micropyrites display a remarkably narrow range of Δpyr (i.e. Δpyr ≡ δ34SSO4 − δ34Spyr) between 56 and 62 ‰. These measured Δpyr values agree with sulfate-sulfide equilibrium fractionation, as observed in natural settings characterised by low microbial sulfate reduction respiration rates. Moreover, the distribution of S isotope compositions recorded in the studied micropyrites suggests that sulfide oxidation also occurred at the microbialite scale. These results highlight the potential of micropyrites to capture signatures of microbial sulfur cycling and show that S isotope composition in pyrites record primarily the local micro-environments induced by the microbialite. 1δ34S = ((34S/32S)sample/(34S/32S)reference − 1) × 1000 in ‰, with Vienna Canyon Diablo Troilite as the reference. |
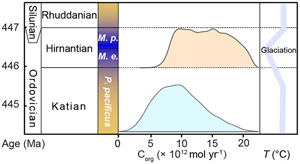 | Enhanced organic carbon burial intensified the end-Ordovician glaciation Abstract: The end-Ordovician (Hirnantian) glaciation, causally linked with the first of five Phanerozoic mass extinction events, is the first major Phanerozoic-glaciation with short duration and paradoxically happened under a relative greenhouse-condition. Here, we present the first zinc isotope study on both carbonate and shale successions that span the Ordovician–Silurian boundary interval in South China. Two positive shifts in Zn isotope composition are observed during two main periods of glacial maxima, indicative of two pulses of extensive carbon burial. The enhanced organic carbon burial during glacial maxima intervals might be causatively linked to cooling-induced elevation in organic carbon burial efficiency. This implies that a large oceanic organic carbon reservoir played a vital role in regulating the atmospheric pCO2, causing the Hirnantian glaciation and producing the pattern of glacial-to-deglacial change, sensitive to the temperature effect. |
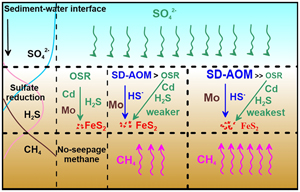 | Enrichment mechanism of trace elements in pyrite under methane seepage Abstract: Authigenic pyrite is an important recorder for methane seepage. During methane seepage, the sulfur and iron isotopic composition of pyrite will change, allowing it to be used as an indicator to identify methane seepage. However, the dissimilar behaviour of trace elements in authigenic pyrite during methane seepage remain unclear. To provide insights, we used pyrite samples from the ‘Haima’ seep locality to determine differences in trace element contents in pyrite obtained from the sulfate-methane transition zone (SMTZ) and that from normal (non-seepage) sedimentary environments. In the SMTZ, the content of cadmium (Cd) related to the organoclastic sulfate reduction in pyrite was low, while the molybdenum (Mo) content, which is highly sensitive to redox environments, was high. This discrepancy can be explained by the fact that sulfate ions (SO42−) in the SMTZ were preferentially consumed by sulfate driven anaerobic oxidation of methane (SD-AOM), which inhibited organoclastic sulfate reduction and decreased the trace metals derived from organic matter in pore water. Simultaneously, intense SD-AOM produced more hydrogen sulfide (H2S), which was more conducive to Mo removal from pore water, and then more Mo adsorption onto pyrite. Further analysis shows that the Mo/Cd ratio of pyrite in the SMTZ (average value of 82.08) is significantly higher than that of the non-SMTZ (average value of 16.02). We believe that the Mo/Cd ratio has great potential to indicate methane seepage, and thus provides a new indicator for methane seepage research. |
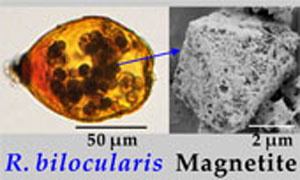 | Magnetic foraminifera thrive in the Mariana Trench Abstract: Unicellular magnetic microorganisms include magnetotactic bacteria and some protist species. Although magnetosome magnetite in bacteria (prokaryotes) is well studied, little is known regarding the characteristics and origin of magnetic minerals in protists (eukaryotes). Stercomata stored within tests of the hadal foraminifera R. bilocularis from the Mariana Trench (6980–10,911 m depth), contain magnetite crystals. As a result, this species can orient in accordance with magnetic fields. The magnetite differs chemically and physically from that in the surrounding sediments. The crystals also differ from bacterial magnetosomes in being of variable size, porous structure, not arranged in chains, and encapsulated in a lipid membrane. Putting available evidence together indicates a biological origin of the magnetite, although a sedimentary source cannot be eliminated. This is the first record of a magnetic protist from hadal depths, opening a new window for the biomagnetism in the Earth’s extreme environment. |
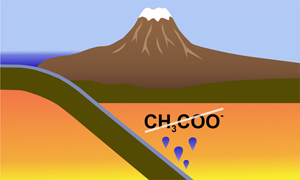 | On the stability of acetate in subduction zone fluids Abstract: Recent theoretical studies have suggested that, under certain intermediate redox conditions, organic anions, in particular acetate, may be the dominant carbon species in deep subduction zone fluids. This could have major consequences for the properties of these fluids, for the formation of diamonds, and for the deep carbon cycle in general. We have tested these predictions by carrying out both ex situ piston cylinder experiments and in situ Raman spectroscopic experiments in the externally heated diamond cell, with a temperature up to 600 °C and pressure up to 5 GPa, the predicted stability field of acetate. We observed that upon heating and pressurisation to these conditions, an aqueous solution of sodium acetate undergoes several interesting reactions, including the formation of higher hydrocarbons. However, at 5 GPa and 600 °C, almost all organic species appear to have decomposed to a graphite-like material. Our experiments, therefore, do not support the stability of acetate and other organics as main carbon species in deep subduction fluids. However, the stability of minor concentrations of such species is still possible and requires further study. During deep subduction, most of the reduced carbon in sediments may be retained and recycled into the mantle to great depth. |
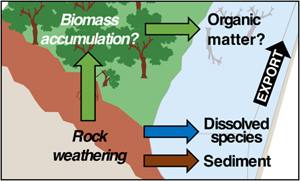 | A global imbalance in potassium and barium river export: the result of biological uptake? Abstract: The role of biological cycling on the chemistry of rivers remains poorly understood. In an attempt to close this knowledge gap, here we examine the difference between the elemental supply to catchments through rock degradation and the corresponding elemental riverine export, for two non-nutrient elements lithium (Li) and sodium (Na) and two nutrients-like elements potassium (K) and barium (Ba), in 20 of the largest world river catchments. Overall, the riverine export of K and Ba are lower than their estimated release by catchment scale rock degradation, while the two fluxes match for Li and Na. Barium isotope constraints lending support to this observation, we take this difference between these two element groups as a suggestion of the influence of biological uptake of rock-derived nutrients on river chemistry. Nevertheless, the magnitude of riverine K depletion cannot be reconciled with a pervasive growth of the biota on continents, nor with an “occult” export of organic material that would go unnoticed by common sampling protocols. One plausible explanation for this conundrum could lie in the complex partitioning of elements amongst soil, biota, and dead organic matter. As a consequence, our study emphasises the need for further work aiming at deciphering the cycle of rock-derived nutrients in the Critical Zone. |
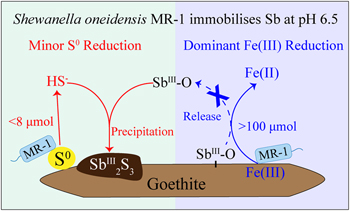 | Iron(III) reducing bacteria immobilise antimonite by respiring elemental sulfur Abstract: Iron(III) reducing bacteria (IRB) are best known and most influential drivers for mobilising metal(loid)s via reductive dissolution of iron(III)-containing minerals. Our study challenges this preconception and found that IRB immobilise metal(loid)s by respiring elemental sulfur (S0) even when Fe(III) reduction is prevailing under mildly acidic conditions. Antimony (Sb), a toxic contaminant, was chosen as an example of metal(loid)s. Antimonite-adsorbed goethite was incubated with Shewanella oneidensis MR-1, a widely distributed IRB, in the presence of S0 at pH 6.5. The results show that although the extent of Fe(III) reduction (>100 μmol) was over ten times greater than that of S0 reduction (<8 μmol), it was S0 reduction that immobilised Sb through Sb2S3 precipitation. Further, the thermodynamic calculation suggests that such great impacts of marginal S0 reduction can be extended to other metal(loid)s via the formation of soluble thio-species or sulfide precipitates. This study redefines the role of IRB in the environmental fate of metal(loid)s, highlighting the strong impacts from the marginal S0 reduction over the central Fe(III) reduction. |
 | Martian core composition from experimental high-pressure metal-silicate phase equilibria Abstract: Current Martian core composition models suggest an iron-rich core alloyed with 10 to 20 wt. % of sulfur. Although Mars is more oxidised than Earth, oxygen is usually discarded as a potential light element candidate, since its dissolution into iron is negligible at the pressures and temperatures prevailing during Mars’s primitive differentiation. However, it has recently been shown that oxygen interacts with the sulfur in the metal, which dramatically increases its solubility. Here, we investigated this novel process by carrying out metal-silicate equilibration experiments between 2 and 12 GPa, and 1673 and 2473 K, using piston-cylinder and multi-anvil presses. The experimental results show that oxygen was systematically incorporated in the metallic phase alongside sulfur, and a thermodynamic model was developed to parametrise this interaction. The oxygen-sulfur interaction parameter arising from those thermodynamic equations was fitted and used in a multi-stage core modelling simulation. We found that a Martian core containing 14 to 19 wt. % S (maximum permissible concentration according to cosmochemical constraints) will also contain between 1.3 and 3.5 wt. % O. This would help to match the Martian core density estimate while being cosmochemically consistent. |
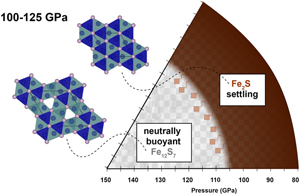 | Stability of Fe2S and Fe12S7 to 125 GPa; implications for S-rich planetary cores Abstract: The Fe-FeS phase relations were explored in the 22–25 wt. % S compositional range using single crystal X-ray diffraction in a laser heated diamond anvil cell. At pressures up to 125 GPa and at high temperatures, Fe2S and Fe12S7 were determined to co-crystallise. The novel Fe12S7 compound adopts the Co12P7 structure and Fe2S assumes the Fe2P-type structure. Applying these results to an Fe-FeS binary phase diagram exposes a complex series of FeS phase assemblages in the 16–25 wt. % S range, whereby minor changes in S content significantly affect the crystallisation sequence of Fe-S rich planetary cores. For core compositions S-rich of the Fe2S-Fe12S7 eutectic, the small density difference between solid Fe12S7 and Fe2S is likely to result in the formation of a core slush rather than a gravitationally stable inner core. Crystallisation of denser Fe2S at eutectic conditions could then result in gravitational settling of an Fe2S-rich inner core over time. As the Fe2P-type Fe2S has previously been identified forming at high temperatures to pressures as low as 22 GPa, the core crystallisation regimes determined here also elucidate that the Martian core sulfur composition must lie on the S-rich side of the Fe-Fe3S eutectic or even the S-rich side of the Fe3S-Fe2S eutectic to maintain a fully molten core. |
<< Previous issueNext issue >>





