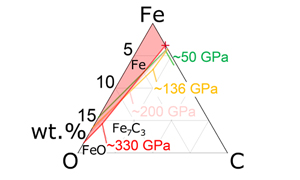
 | Melting experiments on Fe-C-O to 200 GPa; liquidus phase constraints on core composition Abstract: Recent theoretical calculations suggested that carbon and oxygen are important light elements in the Earth’s inner and outer core, respectively. We performed melting experiments on the Fe-C-O system and obtained ternary liquidus phase relations at ∼50, ∼136, and ∼200 GPa based on textural and compositional characterisations of recovered samples. Considering the previously reported Fe-C binary eutectic liquid composition, these results are extrapolated to 330 GPa, which constrains C and O concentrations in the liquid outer core that crystallises Fe at the inner core. Theory has predicted a possible range of the solid inner core composition in Fe-C-S-Si that explains seismological observations. The compositions of liquids Fe-C-O-S-Si in equilibrium with such solid Fe-C-S-Si alloys are calculated with the solid-liquid partition coefficient of C obtained in this study along with those of S and Si in the literature. These liquid compositions, however, do not satisfy constraints from both outer core observations and the liquidus phase relations examined in this study, suggesting that the inner core is not Fe-C-S-Si alloy but may include H as an important impurity element. |
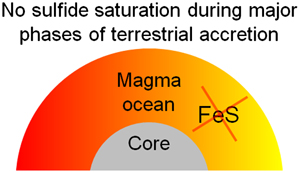 | Sulfur solubility in a deep magma ocean and implications for the deep sulfur cycle Abstract: The Earth could have experienced sulfide segregation during its differentiation due to sulfur (S) saturation within a magma ocean. The relative timing of sulfide saturation during magma ocean crystallisation is strongly dependent on the solubility of S at sulfide saturation (SCSS). Here, we present SCSS data directly relevant for a deep terrestrial magma ocean obtained from laser heated diamond anvil cell experiments. Our new data, along with existing SCSS data obtained for similar compositions, was parameterised to derive a new predictive equation. Our parameterisation predicts that existing models strongly underestimate the SCSS over the P-T range of a deep magma ocean. Our SCSS models provide the S abundances required at any given stage of terrestrial accretion, and imply that sulfide saturation is much less efficient at stripping the Earth’s mantle of S during accretion than previously predicted. Applying our results to the most recent mantle S evolution models shows that the SCSS would be far too high to achieve sulfide saturation, until only perhaps the final stages of magma ocean crystallisation. To satisfy highly siderophile element systematics, either the S content of the magma ocean was considerably higher than currently assumed, or highly siderophile element abundances were affected by other processes, such as iron disproportionation. |
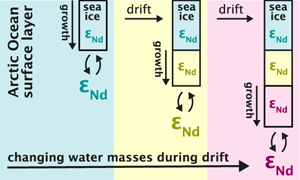 | Neodymium isotopes trace marine provenance of Arctic sea ice Abstract: Radiogenic neodymium (Nd) isotopes (ɛNd) have the potential to serve as a geochemical tracer of the marine origin of Arctic sea ice. This capability results from pronounced ɛNd differences between the distinct marine and riverine sources, which feed the surface waters from which the ice forms. The first dissolved Nd isotope and rare earth element (REE) concentration data obtained from Arctic sea ice collected across the Fram Strait during RV Polarstern cruise PS85 in 2014 confirm the incorporation and preservation of the parental surface seawater ɛNd signatures despite efficient REE rejection. The large ɛNd variability between ice floes and within sea ice cores (−32 to −10) reflects changes in water mass distribution during ice growth and drift from the central Arctic Ocean to Fram Strait. In addition to the parental seawater composition, our new approach facilitates the reconstruction of the transfer of matter between the atmosphere, the sea ice and the ocean. In conjunction with satellite-derived drift trajectories, we enable a more accurate assessment of sea ice origin and spatiotemporal evolution, benefiting studies of sea ice biology, biodiversity, and biogeochemistry. |
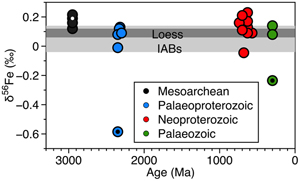 | Constant iron isotope composition of the upper continental crust over the past 3 Gyr Abstract: The Fe isotopic composition of twenty four glacial diamictite composites with depositional ages ranging from the Mesoarchean to the Palaeozoic serve as proxies of the average upper continental crust (UCC) and can be used to track how δ56Fe may have changed in the continental crust through time. The diamictites have elevated chemical index of alteration (CIA) values and other characteristics of weathered regoliths (e.g., strong depletion in soluble elements such as Sr), which they inherited from their upper crustal source regions. The δ56Fe values in the diamictite composites range from −0.59 ‰ to +0.23 ‰. Excluding three samples impacted by the incorporation of materials from Fe formations, the diamictites have an average δ56Fe of 0.12 ± 0.13 ‰ (2σ), overlapping the recent estimated average δ56Fe of 0.09 ± 0.03 ‰ (2 s.d.) in the upper continental crust (Dauphas et al., 2017, and references therein). There is no obvious correlation between δ56Fe of the glacial diamictites and the CIA. Our data suggest that the Fe isotope composition of the upper continental crust has been relatively constant throughout Earth history and that chemical weathering is not important in producing Fe isotope variations in the upper continental crust. Pre-Great Oxidation Event (GOE) anoxic weathering, when iron was soluble in its divalent state, did not generate different Fe isotopic signatures from the post-GOE oxidative weathering environment in the upper continental crust. Therefore, the large Fe isotopic fractionations observed in various marine sedimentary records are likely due to processes occurring in the oceans (e.g., biological activity) rather than abiotic redox reactions on the continents. |
 | How long for plastics to decompose in the deep sea? Abstract: The deep sea floor is recognised as one of the most important final destinations for plastic debris. It is not clear whether the plastic debris in the deep sea could be degraded. Likewise, little is known about how long plastics might last at the deep sea floor. A total of 103 plastic debris were recovered using the manned submersible “Shenhaiyongshi” on the deep sea floor (746–3997 m) of the South China Sea (SCS). We found that abundant corrosion structures were present on the surface of polyethylene (PE), which was the dominant type of plastic sample (80 %). The rod-like, filamentous and peanut-like morphologies of the corrosion structures are well in line with those of microorganisms, suggesting that they were derived from biodegradation. The calculation of volume loss of corroded PE showed that about 1.08–13.72 % PE were degraded. Assuming that the most degraded plastic reached the deep sea floor 40 years ago, these plastics will require about 292 years to be totally degraded. Our results provide unique insights into the fate of deep sea plastics and answer the unsolved question about how long plastics may persist in deep sea. |
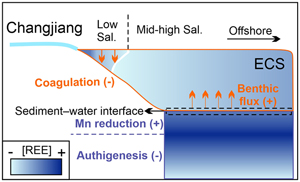 | Dominance of benthic flux of REEs on continental shelves: implications for oceanic budgets Abstract: Rare earth elements (REEs) are powerful tools to track oceanic biogeochemical processes. However, our understanding of REE sources is incomplete, leading to controversial interpretations regarding their oceanic cycling. Continental margin sediments are often assumed to be a major source, but the sediment pore water data required to understand the processes controlling that potential source are scarce. Here, we measure and compile pore water and estuarine REE data from the Changjiang (Yangtze) estuary–East China Sea shelf. We show that release of REEs, from shallow pore water to overlying seawater, is coupled to Mn reduction. In contrast, REEs are removed in deep pore water, perhaps via formation of an authigenic REE-bearing phase. This sedimentary source can potentially explain REE addition in the estuary at mid-high salinity. Our calculations suggest that the benthic flux is the largest Nd source (∼40 %) on the East China Sea shelf. Globally, however, despite a higher benthic Nd flux on the advection-dominated shelf, the much more extensive deep ocean still dominates the total area-integrated benthic flux. Our results call for a more extensive investigation of the magnitude of the benthic flux of REEs to the oceans. |
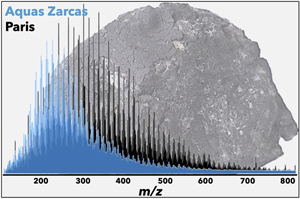 | Diversity of chondritic organic matter probed by ultra-high resolution mass spectrometry Abstract: Primitive carbonaceous chondrites exhibit an unparalleled diversity in terms of their organic content, in addition to a variable degree of hydrothermal alteration. Whether this diversity results from the circulation of fluids or from a multiplicity of precursors remains an open question of prime interest to understand the formation of carbonaceous asteroids. We applied laser desorption ionization Fourier transform ion cyclotron resonance mass spectrometry (LDI-FTICR-MS) on the macromolecular carbon of recent CM carbonaceous chondrite falls, as well as Orgueil (CI) and Tarda (C2). We probed the diversity of molecular fragments released under low power laser beam. The abundance of the chemical families is correlated to the extent of aqueous alteration, which promotes a structural aromatisation. The weakly altered Paris has retained the largest chemical heterogeneity, whilst it is lost in more altered chondrites. Orgueil and Tarda insoluble organic matter share similarities; this is consistent with Tarda and Orgueil originating from the outer belt region. Applied to returned asteroidal samples, FTICR-MS may help unravelling the origin and evolution of organic compounds during the early stages of the solar system. |
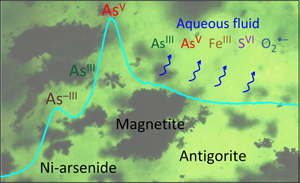 | Redox dynamics of subduction revealed by arsenic in serpentinite Abstract: Redox dynamics of subduction processes remain poorly constrained owing to the lack of direct geochemical tracers. We studied, using X-ray absorption spectroscopy, the chemical and redox state of arsenic in the Tso Morari serpentinites that are witnesses of the Himalayan subduction. Our measurements reveal remarkably contrasting redox speciation, from arsenide (As–III) to arsenite (AsIII) and arsenate (AsV). Combined with physical-chemical constraints, these data enable reconstruction of the ‘redox travel’ of arsenic in the subduction process. Upon early serpentinisation of mantle peridotite, arsenic was scavenged from the fluid and dragged down as insoluble nickel arsenide. Partial deserpentinisation close to the peak metamorphism (550–650 °C) resulted in oxidative dissolution of arsenide to aqueous AsIII and AsV and their non-specific intake by antigorite. The AsV/AsIII ratios (∼0.1–10) analysed in the mineral are ∼104 times higher on average than predicted assuming bulk system thermodynamic equilibrium. These findings reflect a transient out-of-equilibrium release of highly oxidised fluids, with fO2 reaching ∼10 log units above the fayalite-magnetite-quartz buffer (FMQ+10). Arsenic in serpentinite is thus a sensitive record of subduction redox dynamics inaccessible when using traditional equilibrium approaches applied to bulk fluid-mineral systems. |
 | Equilibrium olivine-melt Mg isotopic fractionation explains high δ26Mg values in arc lavas Abstract: We determined equilibrium Mg isotope fractionation between olivine and melt (Δ26/24MgOl/melt) in five, naturally quenched, olivine-glass pairs that were selected to show clear textural and chemical evidence of equilibration. We employed a high-precision, critical mixture double-spiking approach to obtain a weighted mean of Δ26/24MgOl/melt = −0.071 ± 0.010 ‰, for values corrected to a common olivine-glass temperature of 1438 K. As function of temperature, the fractionation can be expressed as Δ26/24MgOl/melt = (−1.46 ± 0.26) × 105/T2. The samples analysed have variable H2O content from 0.1 to ∼1.2 wt. %, yet no discernible difference in Δ26/24MgOl/melt was evident. We have used this Δ26/24MgOl/melt to revisit the puzzling issue of elevated Mg isotope ratios in arc lavas. In new Mg isotope data on sample suites from the Lesser Antilles and Mariana arcs, we show that primitive samples have MORB-like Mg isotope ratios while the evolved samples tend to have isotopically heavier compositions. The magnitude of this variability is well explained by olivine fractionation during magmatic differentiation as calculated with our new equilibrium Δ26/24MgOl/melt. |
 | Corrigendum to “How long for plastics to decompose in the deep sea?” by Zhang and Peng, 2022 |
 | Corrigendum to “Oxygen limitation can trigger the production of branched GDGTs in culture” by Halamka et al., 2021 |
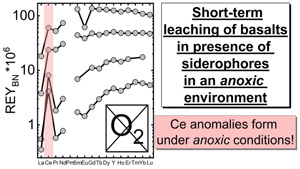 | Siderophores and the formation of cerium anomalies in anoxic environments Abstract: Microbes and plants affect the mobility of trace elements and may have done so since the onset of “life” on Earth. The recognition of such bio-effects on early Earth or Mars remains challenging and the impact of organisms on element mobilisation and redox cycling is largely unconstrained. Redox-sensitive trace elements, such as Ce, are used as geochemical proxies for reconstructing atmospheric oxygen levels, but bio-proxies are still largely lacking. We show that experimental water–rock interaction in presence of siderophores, globally abundant biogenic ligands excreted by microbes, fungi and plants, enhances lanthanoide mobilisation and produces positive Ce anomalies even under strictly anoxic conditions. This is the first evidence for bio-mediated oxidation of Ce and Ce anomaly formation in anoxic environments. Oxygen-independent fractionation of Ce from its redox-insensitive rare earth element neighbours during geo–bio interaction may hold the potential to use Ce anomalies as a bio-proxy in addition to its current use as a redox proxy. |
<< Previous issueNext issue >>





