Metamorphic evolution of carbonate-hosted microbial biosignatures
Affiliations | Corresponding Author | Cite as | Funding information- Share this article





Article views:3,591Cumulative count of HTML views and PDF downloads.
- Download Citation
- Rights & Permissions
Abstract

Figures and Tables
 Figure 1 Starting material for experiments showing (a) Six high pressure, high temperature (HPHT) experimental conditions investigated and their corresponding metamorphic grades spanning lower and upper blueschist (BS), greenschist (GS) and amphibolite (Amph.). Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012; Li et al., 2013b, 2014; Picard et al., 2015a,b; Alleon et al., 2016, 2017; Miot et al., 2017). (b) Si-rich microbial filamentous material and coccoidal cell structures are observed under SEM in the the starting material, shown in (c) by a view ~ 1 cm across; forming Si-rich biofilms around calcite grains (Fig. S-1a,b). | 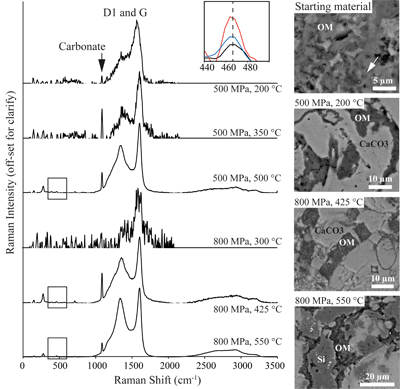 Figure 2 Evolution of D1 (~1360 cm-1) and G (~1610 cm-1) Raman peaks from experimental samples, and the increasing 467 cm-1 quartz peak at 425, 500, and 550 °C (box inset); and corresponding SEM images showing the textural evolution of OM mineralisation within the carbonate (CaCO3) matrix and siliceous (Si) phases Individual cells in the amorphous organic matrix can be seen in the starting material (arrows). Unprocessed Raman spectra are given in Figure S-3. |  Figure 3 BSE+EDS elemental maps or plain BSE image (left), where red = Ca and green = Si, and corresponding Raman maps (right) of the same region, where fuchsia = calcite (1006 cm-1 band), orange/yellow = quartz (465 cm-1 band), green = carbon (combined D1 and G bands), light pink = resin, black = masked fluorescence. (a) Starting material; (b) 800 MPa, 300 °C; (c) 500 MPa, 200 °C; (d) 800 MPa, 425 °C; (e) 500 MPa, 350 °C; (f) 800 MPa, 550 °C; (g) 500 MPa, 500 °C; (h) 800 MPa, 550 °C. EDS spectra are provided in Figure S-5. |  Figure 4 (a – g) BSE+EDS elemental maps (red = Ca, green = Si, yellow = S) and SEM images of sulfur globules in (a, b) Starting material; (c) 800 MPa, 425 °C; (d, e) 800 MPa, 300 °C; (f) 800 MPa, 550 °C; and (g) 500 MPa, 500 °C; (h) BSE+EDS elemental map showing the co-location of Fe (Fe = purple) with S (S = yellow) within a silica-rich (Si = red) fabric (i) in the 800 MPa, 425 °C experiment. |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 |
top
Introduction
Our knowledge of microbiological evolution is informed by organic, morphological, and geochemical biosignatures preserved within a rock record that is destroyed or modified over geological time (Westall, 2008
Westall, F. (2008) Morphological Biosignatures in Early Terrestrial and Extraterrestrial Materials. Space Science Reviews 135, 95–114.
). Chemically-precipitated sediments, particularly carbonate and silica, host microfossils spanning much of Earth history, including in association with early evidence for microbial life on Earth (Moreau and Sharp, 2004Moreau, J.W., Sharp, T.G. (2004) A Transmission Electron Microscopy Study of Silica and Kerogen Biosignatures in ~1.9 Ga Gunflint Microfossils. Astrobiology 4, 196–210.
; Westall, 2008Westall, F. (2008) Morphological Biosignatures in Early Terrestrial and Extraterrestrial Materials. Space Science Reviews 135, 95–114.
; Wacey et al., 2011Wacey, D., Kilburn, M.R., Saunders, M., Cliff, J., Brasier, M.D. (2011) Microfossils of sulphur-metabolizing cells in 3.4-billion-year-old rocks of Western Australia. Nature Geoscience 4, 698–702.
; Djokic et al., 2017Djokic, T., Van Kranendonk, M., Campbell, K.A., Walter, M.R., Ward, C.R. (2017) Earliest signs of life on land preserved in ca. 3.5 Ga hot spring deposits. Nature Communications 8, 15263, doi: 10.1038/ncomms15263.
). The petrographic relationships between microbially-derived kerogen and the inorganic matrix are important in establishing microfossil biogenicity (Brasier et al., 2005Brasier, M.D., Green, O.R., Lindsay, J.F., McLoughlin, N., Steele, A., Stoakes, C. (2005) Critical testing of Earth’s oldest putative fossil assemblage from the ∼3.5 Ga Apex chert, Chinaman Creek, Western Australia. Precambrian Research 140, 55–102.
; Wacey et al., 2011Wacey, D., Kilburn, M.R., Saunders, M., Cliff, J., Brasier, M.D. (2011) Microfossils of sulphur-metabolizing cells in 3.4-billion-year-old rocks of Western Australia. Nature Geoscience 4, 698–702.
; Foucher and Westall, 2013Foucher, F., Westall, F. (2013) Raman Imaging of Metastable Opal in Carbonaceous Microfossils of the 700–800 Ma Old Draken Formation. Astrobiology 13, 57–67.
), in addition to understanding broader interations of microbe-mineral systems back into the early Archean (Westall et al., 2015Westall, F., Campbell, K.A., Bréhéret, J.G., Foucher, F., Gautret, P., Hubert, A., Sorieul, S., Grassineau, N., Guido, D.M. (2015) Archean (3.33 Ga) microbe-sediment systems were diverse and flourished in a hydrothermal context. Geology 43, 615–618.
).Significant experimental effort has furthered our understanding of microbial biomineralisation (Westall et al., 1995
Westall, F., Boni, L., Guerzoni, E. (1995) The experimental silicification of microorganisms. Palaeontology 38, 495–528.
; Orange et al., 2012Orange, F., Disnar, J.R., Gautret, P., Westall, F., Bienvenu, N., Lottier, N., Prieur, D. (2012) Preservation and evolution of organic matter during experimental fossilisation of the hyperthermophilic archaea Methanocaldococcus jannaschii. Origins of Life and Evolution of Biospheres 42, 587–609.
, 2014Orange, F., Dupont, S., Goff, O.L., Bienvenu, N., Disnar, J.R., Westall, F., Le Romancer, M., (2014) Experimental fossilization of the thermophilic Gram-positive Bacterium Geobacillus SP7A: a long duration preservation study. Geomicrobiology Journal, 31, 578–589.
; Li et al., 2013aLi, J., Benzerara, K., Bernard, S., Beyssac, O. (2013a) The link between biomineralization and fossilization of bacteria: Insights from field and experimental studies. Chemical Geology 359, 49–69.
and references therein; Gaboyer et al., 2017Gaboyer, F., Milbeau, C.L., Bohmeier, M., Schwendner, P., Vannier, P., Beblo-Vranesevic, K., Rabbow, E., Foucher, F., Gautret, P., Guégan, R., Richard, A., Sauldubois, A., Richmann, P., Perras, A.K., Moissl-Eichinger, C., Cockell, C.S., Rettberg, P., Marteinsson, V., Monaghan, E., Ehrenfreund, P., Garcia-Descalzo, L., Gomez, F., Malki, M., Amils, R., Cabezas, P., Walter, N., Westall, F. (2017) Mineralization and Preservation of an extremotolerant Bacterium Isolated from an Early Mars Analog Environment. Scientific Reports 7, 14.
). High pressure, high temperature (HPHT) experiments are now shedding light on microfossil taphonomy during geological sequestration, with a focus on thermal degradation of biogenic minerals (Li et al., 2013bLi, Y.L., Konhauser, K.O., Kappler, A., Hao, X.L. (2013b) Experimental low-grade alteration of biogenic magnetite indicates microbial involvement in generation of banded iron formations. Earth and Planetary Science Letters, 361, 229–237.
) and biomineralised cells (Li et al., 2014Li, J., Bernard, S., Benzerara, K., Beyssac, O., Allard, T., Cosmidis, J., Moussou, J. (2014) Impact of biomineralization on the preservation of microorganisms during fossilization: An experimental perspective. Earth and Planetary Science Letters 400, 113–122.
). Experiments have demonstrated the persistence of microbiological structures such as iron oxide organominerals up to 250 °C and 140 MPa (Picard et al., 2015aPicard, A., Kappler, A., Schmid, G., Quaroni, L., Obst, M. (2015a) Experimental diagenesis of organo-mineral structures formed by microaerophilic Fe(II)-oxidizing bacteria. Nature Communications 6, 6277 (2015), doi: 10.1038/ncomms7277.
), and the preservation of lipids and polysaccharides within Fe-encrusted microbial cells at the same PT conditions (Picard et al., 2015bPicard, A., Obst, M., Schmid, G., Zeitvogel, F., Kappler, A. (2015b) Limited influence of Si on the preservation of Fe mineral-encrusted microbial cells during experimental diagenesis Geobiology 14, 276–292.
). Recently, Alleon et al. (2016)Alleon, J., Bernard, S., Le Guillou, C., Daval, D., Skouri-Panet, F., Pont, S., Delbes, L., Robert, F. (2016) Early entombment within silica minimizes the molecular degradation of microorganisms during advanced diagenesis. Chemical Geology 437, 98–108.
showed that microbial entombment within silica limited the degradation of molecular biosignatures exposed to 25 MPa and 250 °C for 100 days.As improving analytical techniques expand biosignature studies into increasingly metamorphosed terrains (Bernard et al., 2007
Bernard, S., Benzerara, K., Beyssac, O., Mgnguy, N., Guyot, F., Brown Jr., G.E., Goffe, B. (2007) Exceptional preservation of fossil plant spores in high-pressure metamorphic rocks. Earth and Planetary Science Letters 262, 257–272
, 2010Bernard, S., Benzerara, K., Beyssac, O., Brown, G.E. (2010) Multiscale characterization of pyritized plant tissues in blueschist facies metamorphic rocks. Geochimica Cosmochimica Acta 74, 5054–5068.
; Galvez et al., 2012Galvez, M.W., Beyssac, O., Benzerra, K., Bernard, S., Menguy, N., Cox, C.S., Martinez, I., Johnston, M.R., Brown Jr, G.E. (2012) Morphological preservation of carbonaceous plant fossils in blueschist metamorphic rocks from New Zealand. Geobiology 10, 118–129.
; Papineau et al., 2019Papineau, D., DeGregorio, B., Sagar, J., Thorogate, R., Wang, J., Nittler, L., Kilcoyne, D.A., Marbach, H., Drost, M. Thornton, G. (2019) Fossil biomass preserved as graphitic carbon in a late Paleoproterozoic banded iron formation metamorphosed at more than 550 ºC. Journal of the Geological Society of London, jgs2018-097.
), there is a need to understand the influence of higher PT regimes (Li et al., 2013aLi, J., Benzerara, K., Bernard, S., Beyssac, O. (2013a) The link between biomineralization and fossilization of bacteria: Insights from field and experimental studies. Chemical Geology 359, 49–69.
). We present an experimental investigation into the effects of HPHT on carbonate-hosted microbial biofilms from a natural thermal spring environment. We trace the first stages of petrographic evolution of geochemical and morphological sample components in response to 500 and 800 MPa and temperatures spanning 200 to 550 °C, significantly expanding the PT space previously investigated (Fig. 1a).top
Sample Material
Porous carbonate precipitates containing green, orange, and grey chasmolithic microbial communities (Fig. 1c) were collected from a CO2 thermal spring (Jotun Spring; Banks et al., 1998
Banks, D., Sletten, R.S., Haldorsen, S., Dale, B., Heim, M., Swensen, B. (1998) The Thermal Springs of Bockfjord, Svalbard: Occurrence and Major Ion Hydrochemistry. Geothermics 27, 445–467.
) in Spitzbergen, Svalbard, during the Arctic Mars Analog Svalbard Expeditions from 2006-2011 (Starke et al., 2013Starke, V., Kirshtein, J., Fogel, M.L., Steele, A. (2013) Microbial community composition and endolith colonization at an Arctic thermal spring are driven by calcite precipitation. Environmental Microbiology Reports 5, 648-659, doi: 10.1111/1758-2229.12063.
). An air dried sample was subsampled into multiple ~2 mm3 fragments for six HPHT experiments. Four additional fragments of this starting material were subjected to the same preparation and analytical techniques. Methods are described in the Supplementary Information.
Figure 1 Starting material for experiments showing (a) Six high pressure, high temperature (HPHT) experimental conditions investigated and their corresponding metamorphic grades spanning lower and upper blueschist (BS), greenschist (GS) and amphibolite (Amph.). Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012
Schiffbauer, J. D., Wallace, A.F., Hunter, J.L., Kowalewski, M., Bodnar, R.J., Xiao, S. (2012) Thermally‐induced structural and chemical alteration of organic‐walled microfossils: an experimental approach to understanding fossil preservation in metasediments. Geobiology 10, 402–423.
; Li et al., 2013bLi, Y.L., Konhauser, K.O., Kappler, A., Hao, X.L. (2013b) Experimental low-grade alteration of biogenic magnetite indicates microbial involvement in generation of banded iron formations. Earth and Planetary Science Letters, 361, 229–237.
, 2014Li, J., Bernard, S., Benzerara, K., Beyssac, O., Allard, T., Cosmidis, J., Moussou, J. (2014) Impact of biomineralization on the preservation of microorganisms during fossilization: An experimental perspective. Earth and Planetary Science Letters 400, 113–122.
; Picard et al., 2015aPicard, A., Kappler, A., Schmid, G., Quaroni, L., Obst, M. (2015a) Experimental diagenesis of organo-mineral structures formed by microaerophilic Fe(II)-oxidizing bacteria. Nature Communications 6, 6277 (2015), doi: 10.1038/ncomms7277.
,bPicard, A., Obst, M., Schmid, G., Zeitvogel, F., Kappler, A. (2015b) Limited influence of Si on the preservation of Fe mineral-encrusted microbial cells during experimental diagenesis Geobiology 14, 276–292.
; Alleon et al., 2016Alleon, J., Bernard, S., Le Guillou, C., Daval, D., Skouri-Panet, F., Pont, S., Delbes, L., Robert, F. (2016) Early entombment within silica minimizes the molecular degradation of microorganisms during advanced diagenesis. Chemical Geology 437, 98–108.
, 2017Alleon, J., Bernard, S., Le Guillou, C., Daval, D., Skouri-Panet, F., Kuga, M., Robert, F. (2017) Organic molecular heterogeneities can withstand diagenesis. Scientific Reports 7, 1508.
; Miot et al., 2017Miot, J., Bernard, S., Bourreau, M., Guyot, F., Kish, A. (2017) Experimental maturation of Archaea encrusted by Fe-phosphates. Scientific Reports 7, 16984.
). (b) Si-rich microbial filamentous material and coccoidal cell structures are observed under SEM in the the starting material, shown in (c) by a view ~ 1 cm across; forming Si-rich biofilms around calcite grains (Fig. S-1a,b).top
Results
Visible light microscopy of polished HPHT samples reveal dark carbonised organic matter (OM) forming a compositional fabric within the calcite matrix, absent in the starting material (Fig. S-1a,c,d). Secondary electron imaging of a duplicate, acid etched 500 MPa, 350 °C sample shows mineralised biomass maintains its intact extracellular filamentous structure (Figs. 1b and S-1e) and coccoidal cell morphologies (Fig. S-1f). Intact diatom frustules are preserved at the lower PT conditions (500 MPa, 200 °C and 500 MPa, 350 °C), becoming structurally disintegrated in the higher PT experiments (500 MPa, 500 °C; 800 MPa, 300 °C and 800 MPa, 425 °C; Fig. S-1g,h).
Raman spectra of experimental sample surfaces exhibit peaks for carbonate (1100 cm-1), and D1 (1360 cm-1) and G (1610 cm-1) carbon bands (Fig. 2; Pimenta et al., 2007
Pimenta, M.A., Dresselhaus, G., Dresselhaus, M.S., Cancado, L.G., Jorio, A., Saito, R. (2007) Studying disorder in graphite-based systems by Raman spectroscopy. Physical Chemistry Chemical Physics 9, 1276–1291.
). An additional quartz band at 465 cm-1 is observed for the three highest temperature samples (425, 500, and 550 °C), irrespective of pressure. Secondary electron imaging shows OM transitioning from amorphous biogenic material with cellular structures in the starting material to increasingly crystalline structures as PT increases (Fig. 2). This is reflected in the changing carbon G-band centre and width, whereby temperatures of 350 °C and above produce a sharpened (more crystalline) G-band, and the increase in temperature from 200 to 550 °C also produces a peak centre shift to longer wavenumbers (Fig. S-2). Across all HPHT conditions OM maintains its original petrographic texture, either within a biofilm structure (Fig. 1c), or bound within diatom frustules (Fig. 1).
Figure 2 Evolution of D1 (~1360 cm-1) and G (~1610 cm-1) Raman peaks from experimental samples, and the increasing 467 cm-1 quartz peak at 425, 500, and 550 °C (box inset); and corresponding SEM images showing the textural evolution of OM mineralisation within the carbonate (CaCO3) matrix and siliceous (Si) phases Individual cells in the amorphous organic matrix can be seen in the starting material (arrows). Unprocessed Raman spectra are given in Figure S-3.
Raman spectroscopy and SEM+EDS elemental mapping of experimental sample surfaces show kerogenous material (D1 and G Raman bands) remains spatially-concurrent with the siliceous phase (Fig. 3). At the lowest PT condition (500 MPa, 200 °C), petrographic textures are largely indistinguishable from those in the starting material. At 800 MPa, 425 °C (Fig. 3d) these phases form compositional fabrics, where the organic-rich phase exists within silicified or quartz-rich fabrics, which also have elevated Fe (Fig. 4h,i). With increasing PT, the silica-organic phase becomes partitioned from the recrystallising calcite, eventually forming discrete petrographic end members at 800 MPa and 550 °C, whereby kerogen either forms an organic carbonaceous film around quartz crystals (Fig. 3e) or is captured within the quartz itself (Fig. 3f). Water degassing is observed in the carbonate matrix for samples treated at 500 MPa, 350 °C; 500 MPa, 500 °C and 800 MPa, 425 °C (Fig. S-4b,c,e), and extensive fracturing (Fig. S-4b,c,d) is observed at 500 MPa (350 °C and 500 °C) and 800 MPa (300 °C).

Figure 3 BSE+EDS elemental maps or plain BSE image (left), where red = Ca and green = Si, and corresponding Raman maps (right) of the same region, where fuchsia = calcite (1006 cm-1 band), orange/yellow = quartz (465 cm-1 band), green = carbon (combined D1 and G bands), light pink = resin, black = masked fluorescence. (a) Starting material; (b) 800 MPa, 300 °C; (c) 500 MPa, 200 °C; (d) 800 MPa, 425 °C; (e) 500 MPa, 350 °C; (f) 800 MPa, 550 °C; (g) 500 MPa, 500 °C; (h) 800 MPa, 550 °C. EDS spectra are provided in Figure S-5.
Finally, discrete clusters of sulfur globules 1 – 3 μm in size are observed within the starting material (Fig. 4a,b). These globules persist through all thermal conditions at 800 MPa, and at 500 MPa, 500 °C, remaining petrographically associated with the siliceous kerogen-bearing phase (Fig. 4a-g). At the highest PT condition (800 MPa, 550 °C), globules become irregular structures (Fig. 4f). Fe co-occurs within the Si phase for all samples, including in association with the S globules (Fig. 4h). Despite being a strong Raman scatterer, elemental sulfur was not detected by Raman spectroscopy (Fig. 2).

Figure 4 (a – g) BSE+EDS elemental maps (red = Ca, green = Si, yellow = S) and SEM images of sulfur globules in (a, b) Starting material; (c) 800 MPa, 425 °C; (d, e) 800 MPa, 300 °C; (f) 800 MPa, 550 °C; and (g) 500 MPa, 500 °C; (h) BSE+EDS elemental map showing the co-location of Fe (Fe = purple) with S (S = yellow) within a silica-rich (Si = red) fabric (i) in the 800 MPa, 425 °C experiment.
top
Discussion
Within the bounds of a closed system, mm- to micron-scale elemental (Si, S, Fe) biosignatures and microbially-derived kerogen maintain their petrographic relationship throughout initial exposure to HPHT (Fig. S-1c). In situ silicification of biomass occurs without petrographic disruption to the captured OM. With increasing PT, the organic-rich amorphous siliceous phase crystallises into α-quartz, and the originally porous calcite anneals into a crystalline matrix at 800 MPa, 550 °C, expelling siliceous OM-bearing material. Extensive fracturing of the carbonate matrix observed at 800 MPa, 300 °C may be detrimental to preservation of biosignatures in comparison to an annealed calcite matrix, as the fractures would provide pathways for altering fluids. Disintegration of diatom frustules may be an additional source of Si as PT conditions increase.
Displacement of kerogen during diagenetic coarsening of quartz has been identified as a mechanism to explain petrographic relationships in the microfossil-bearing Gunflint chert (Moreau and Sharp, 2004
Moreau, J.W., Sharp, T.G. (2004) A Transmission Electron Microscopy Study of Silica and Kerogen Biosignatures in ~1.9 Ga Gunflint Microfossils. Astrobiology 4, 196–210.
; Foucher and Westall, 2013Foucher, F., Westall, F. (2013) Raman Imaging of Metastable Opal in Carbonaceous Microfossils of the 700–800 Ma Old Draken Formation. Astrobiology 13, 57–67.
). This has implications for the longevity of biosignatures, whereby the quartz fraction resists weathering or secondary aqueous alteration, protecting captured OM (Toporski et al., 2002Toporski, J.K., Steele, A., Westall, F., Thomas-Keprta, K.L., McKay, D.S. (2002) The simulated silicification of bacteria - new clues to the modes and timing of bacterial preservation and implications for the search for extraterrestrial microfossils, Astrobiology 2, 1–26.
). The preservation of OM within silicious material is consistent with Alleon et al. (2016)Alleon, J., Bernard, S., Le Guillou, C., Daval, D., Skouri-Panet, F., Pont, S., Delbes, L., Robert, F. (2016) Early entombment within silica minimizes the molecular degradation of microorganisms during advanced diagenesis. Chemical Geology 437, 98–108.
, who demonstrated the role of silica in limiting thermally-induced molecular degradation of OM in experimental samples. The transformation of Fe minerals to more stable, crystalline phases has also been shown to be conducive to the preservation of organic components (Ferris et al., 1988Ferris, F.G., Fyfe, W.S., Beveridge, T.J. (1988) Metallic ion binding by Bacillus subtilis: implications for the fossilization of microorganisms. Geology 16, 153–157.
; Picard et al., 2015aPicard, A., Kappler, A., Schmid, G., Quaroni, L., Obst, M. (2015a) Experimental diagenesis of organo-mineral structures formed by microaerophilic Fe(II)-oxidizing bacteria. Nature Communications 6, 6277 (2015), doi: 10.1038/ncomms7277.
). Finally, the sulfur globules observed in the starting material and experimental samples are consistent in size and shape to intracellular sulfur globules found within filamentous sulfide oxidising bacteria (Dahl and Prange, 2006Dahl, C., Prange, A. (2006) Bacterial Sulfur Globules: Occurrence, Structure and Metabolism. In: Shively J.M. (Eds.) Inclusions in Prokaryotes. Springer, Berlin, Heidelberg, 21–51.
). Their preservation at the highest HPHT condition holds promise for establishing the biogenicity of similar features in filmentous microfossils (Wacey et al., 2011Wacey, D., Kilburn, M.R., Saunders, M., Cliff, J., Brasier, M.D. (2011) Microfossils of sulphur-metabolizing cells in 3.4-billion-year-old rocks of Western Australia. Nature Geoscience 4, 698–702.
; Bailey et al., 2013Bailey, J.V., Corsetti, F.A., Greene, S.E., Crosby, C.H., Liu, P., Orphan, V.J. (2013) Filamentous sulfur bacteria preserved in modern and ancient phosphatic sediments: implications for the role of oxygen and bacteria in phosphogenesis. Geobiology 11, 397–405.
) and distinguishing them from abiotic S microstructures (Cosmidis and Templeton, 2016Cosmidis, J., Templeton, A.S. (2016) Self-assembly of biomorphic carbon/sulfur microstructures in sulfidic environments. Nature Communications 7, 12812.
).While the OM Raman spectra are relatively close to those observed in the Gunflint and Draken formation (Foucher et al., 2015
Foucher, F., Ammar, M.R., Westall, F. (2015) Revealing the biotic origin of silicified Precambrian carbonaceous microstructures using Raman spectroscopic mapping, a potential method for the detection of microfossils on Mars. Journal of Raman Spectroscopy 46, 873-879.
), such experiments are inheritently limited by their short duration. Even with the addition of P, Raman D1 and G bands of experiment sample OM are broader than those from metasediments of equivalent metamorphic grade (Beyssac et al., 2002Beyssac, O., Goffé, B, Chopin, C., Rouzaud, J.N. (2002) Raman spectra of carbonaceous material in metasediments: a new geothermometer. Journal of Metamorphic Geology 20, 859–871.
), indicating they are less mature. This limitation was also observed by Li et al. (2014)Li, J., Bernard, S., Benzerara, K., Beyssac, O., Allard, T., Cosmidis, J., Moussou, J. (2014) Impact of biomineralization on the preservation of microorganisms during fossilization: An experimental perspective. Earth and Planetary Science Letters 400, 113–122.
for experiments at similar temperatures (300 ºC, 600 ºC) in the absence of pressure.top
Conclusions
This study presents an experimental investigation into the first stages of HPHT evolution of microbial biosignatures within a natural siliceous-carbonate matrix. While thermal maturation of OM is well understood (Vandenbroucke and Largeau, 2007), we show that the early effects of temperature and pressure play an important role in the petrographic redistribution and capture of OM due to the transformation of the low temperature silica and carbonate phases. We demonstrate that exposure up to 800 MPa and 550 °C does not disrupt the petrographic relationship of captured microbial material with its inorganic host phase during the first stages of metamorphism. Future experimental work should address open system effects, including the influence of fluids on the modification of sample components, and longer duration experiments.
top
Acknowledgements
This work was funded by a Royal Society of Edinburgh Research Fellowship. FF and FW acknowledge funding from the CNRS and CNES.
Editor: Karim Benzerara
top
References
Alleon, J., Bernard, S., Le Guillou, C., Daval, D., Skouri-Panet, F., Pont, S., Delbes, L., Robert, F. (2016) Early entombment within silica minimizes the molecular degradation of microorganisms during advanced diagenesis. Chemical Geology 437, 98–108.
 Show in context
Show in context Recently, Alleon et al. (2016) showed that microbial entombment within silica limited the degradation of molecular biosignatures exposed to 25 MPa and 250 °C for 100 days.
View in article
Figure 1 [...] Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012; Li et al., 2013b, 2014; Picard et al., 2015a,b; Alleon et al., 2016, 2017; Miot et al., 2017).
View in article
The preservation of OM within silicious material is consistent with Alleon et al. (2016), who demonstrated the role of silica in limiting thermally-induced molecular degradation of OM in experimental samples.
View in article
Alleon, J., Bernard, S., Le Guillou, C., Daval, D., Skouri-Panet, F., Kuga, M., Robert, F. (2017) Organic molecular heterogeneities can withstand diagenesis. Scientific Reports 7, 1508.
 Show in context
Show in context Figure 1 [...] Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012; Li et al., 2013b, 2014; Picard et al., 2015a,b; Alleon et al., 2016, 2017; Miot et al., 2017).
View in article
Bailey, J.V., Corsetti, F.A., Greene, S.E., Crosby, C.H., Liu, P., Orphan, V.J. (2013) Filamentous sulfur bacteria preserved in modern and ancient phosphatic sediments: implications for the role of oxygen and bacteria in phosphogenesis. Geobiology 11, 397–405.
 Show in context
Show in context Their preservation at the highest HPHT condition holds promise for establishing the biogenicity of similar features in filmentous microfossils (Wacey et al., 2011; Bailey et al., 2013) and distinguishing them from abiotic S microstructures (Cosmidis and Templeton, 2016).
View in article
Banks, D., Sletten, R.S., Haldorsen, S., Dale, B., Heim, M., Swensen, B. (1998) The Thermal Springs of Bockfjord, Svalbard: Occurrence and Major Ion Hydrochemistry. Geothermics 27, 445–467.
 Show in context
Show in context Porous carbonate precipitates containing green, orange, and grey chasmolithic microbial communities (Fig. 1c) were collected from a CO2 thermal spring (Jotun Spring; Banks et al., 1998) in Spitzbergen, Svalbard, during the Arctic Mars Analog Svalbard Expeditions from 2006-2011 (Starke et al., 2013).
View in article
Bernard, S., Benzerara, K., Beyssac, O., Mgnguy, N., Guyot, F., Brown Jr., G.E., Goffe, B. (2007) Exceptional preservation of fossil plant spores in high-pressure metamorphic rocks. Earth and Planetary Science Letters 262, 257–272
 Show in context
Show in context As improving analytical techniques expand biosignature studies into increasingly metamorphosed terrains (Bernard et al., 2007, 2010; Galvez et al., 2012; Papineau et al., 2019), there is a need to understand the influence of higher PT regimes (Li et al., 2013a).
View in article
Bernard, S., Benzerara, K., Beyssac, O., Brown, G.E. (2010) Multiscale characterization of pyritized plant tissues in blueschist facies metamorphic rocks. Geochimica Cosmochimica Acta 74, 5054–5068.
 Show in context
Show in context As improving analytical techniques expand biosignature studies into increasingly metamorphosed terrains (Bernard et al., 2007, 2010; Galvez et al., 2012; Papineau et al., 2019), there is a need to understand the influence of higher PT regimes (Li et al., 2013a).
View in article
Beyssac, O., Goffé, B, Chopin, C., Rouzaud, J.N. (2002) Raman spectra of carbonaceous material in metasediments: a new geothermometer. Journal of Metamorphic Geology 20, 859–871.
 Show in context
Show in context Even with the addition of P, Raman D1 and G bands of experiment sample OM are broader than those from metasediments of equivalent metamorphic grade (Beyssac et al., 2002), indicating they are less mature.
View in article
Brasier, M.D., Green, O.R., Lindsay, J.F., McLoughlin, N., Steele, A., Stoakes, C. (2005) Critical testing of Earth’s oldest putative fossil assemblage from the ∼3.5 Ga Apex chert, Chinaman Creek, Western Australia. Precambrian Research 140, 55–102.
 Show in context
Show in context The petrographic relationships between microbially-derived kerogen and the inorganic matrix are important in establishing microfossil biogenicity (Brasier et al., 2005; Wacey et al., 2011; Foucher and Westall, 2013), in addition to understanding broader interations of microbe-mineral systems back into the early Archean (Westall et al., 2015).
View in article
Cosmidis, J., Templeton, A.S. (2016) Self-assembly of biomorphic carbon/sulfur microstructures in sulfidic environments. Nature Communications 7, 12812.
 Show in context
Show in context Their preservation at the highest HPHT condition holds promise for establishing the biogenicity of similar features in filmentous microfossils (Wacey et al., 2011; Bailey et al., 2013) and distinguishing them from abiotic S microstructures (Cosmidis and Templeton, 2016).
View in article
Dahl, C., Prange, A. (2006) Bacterial Sulfur Globules: Occurrence, Structure and Metabolism. In: Shively J.M. (Eds.) Inclusions in Prokaryotes. Springer, Berlin, Heidelberg, 21–51.
 Show in context
Show in context Finally, the sulfur globules observed in the starting material and experimental samples are consistent in size and shape to intracellular sulfur globules found within filamentous sulfide oxidising bacteria (Dahl and Prange, 2006).
View in article
Djokic, T., Van Kranendonk, M., Campbell, K.A., Walter, M.R., Ward, C.R. (2017) Earliest signs of life on land preserved in ca. 3.5 Ga hot spring deposits. Nature Communications 8, 15263, doi: 10.1038/ncomms15263.
 Show in context
Show in context Chemically-precipitated sediments, particularly carbonate and silica, host microfossils spanning much of Earth history, including in association with early evidence for microbial life on Earth (Moreau and Sharp, 2004; Westall, 2008; Wacey et al., 2011; Djokic et al., 2017).
View in article
Ferris, F.G., Fyfe, W.S., Beveridge, T.J. (1988) Metallic ion binding by Bacillus subtilis: implications for the fossilization of microorganisms. Geology 16, 153–157.
 Show in context
Show in context The transformation of Fe minerals to more stable, crystalline phases has also been shown to be conducive to the preservation of organic components (Ferris et al., 1988; Picard et al., 2015a).
View in article
Foucher, F., Westall, F. (2013) Raman Imaging of Metastable Opal in Carbonaceous Microfossils of the 700–800 Ma Old Draken Formation. Astrobiology 13, 57–67.
 Show in context
Show in context The petrographic relationships between microbially-derived kerogen and the inorganic matrix are important in establishing microfossil biogenicity (Brasier et al., 2005; Wacey et al., 2011; Foucher and Westall, 2013), in addition to understanding broader interations of microbe-mineral systems back into the early Archean (Westall et al., 2015).
View in article
Displacement of kerogen during diagenetic coarsening of quartz has been identified as a mechanism to explain petrographic relationships in the microfossil-bearing Gunflint chert (Moreau and Sharp, 2004; Foucher and Westall, 2013).
View in article
Foucher, F., Ammar, M.R., Westall, F. (2015) Revealing the biotic origin of silicified Precambrian carbonaceous microstructures using Raman spectroscopic mapping, a potential method for the detection of microfossils on Mars. Journal of Raman Spectroscopy 46, 873-879.
 Show in context
Show in context While the OM Raman spectra are relatively close to those observed in the Gunflint and Draken formation (Foucher et al., 2015), such experiments are inheritently limited by their short duration.
View in article
Gaboyer, F., Milbeau, C.L., Bohmeier, M., Schwendner, P., Vannier, P., Beblo-Vranesevic, K., Rabbow, E., Foucher, F., Gautret, P., Guégan, R., Richard, A., Sauldubois, A., Richmann, P., Perras, A.K., Moissl-Eichinger, C., Cockell, C.S., Rettberg, P., Marteinsson, V., Monaghan, E., Ehrenfreund, P., Garcia-Descalzo, L., Gomez, F., Malki, M., Amils, R., Cabezas, P., Walter, N., Westall, F. (2017) Mineralization and Preservation of an extremotolerant Bacterium Isolated from an Early Mars Analog Environment. Scientific Reports 7, 14.
 Show in context
Show in context Significant experimental effort has furthered our understanding of microbial biomineralisation (Westall et al., 1995; Orange et al., 2012, 2014; Li et al., 2013a and references therein; Gaboyer et al., 2017).
View in article
Galvez, M.W., Beyssac, O., Benzerra, K., Bernard, S., Menguy, N., Cox, C.S., Martinez, I., Johnston, M.R., Brown Jr, G.E. (2012) Morphological preservation of carbonaceous plant fossils in blueschist metamorphic rocks from New Zealand. Geobiology 10, 118–129.
 Show in context
Show in context As improving analytical techniques expand biosignature studies into increasingly metamorphosed terrains (Bernard et al., 2007, 2010; Galvez et al., 2012; Papineau et al., 2019), there is a need to understand the influence of higher PT regimes (Li et al., 2013a).
View in article
Li, J., Benzerara, K., Bernard, S., Beyssac, O. (2013a) The link between biomineralization and fossilization of bacteria: Insights from field and experimental studies. Chemical Geology 359, 49–69.
 Show in context
Show in context Significant experimental effort has furthered our understanding of microbial biomineralisation (Westall et al., 1995; Orange et al., 2012, 2014; Li et al., 2013a and references therein; Gaboyer et al., 2017).
View in article
As improving analytical techniques expand biosignature studies into increasingly metamorphosed terrains (Bernard et al., 2007, 2010; Galvez et al., 2012; Papineau et al., 2019), there is a need to understand the influence of higher PT regimes (Li et al., 2013a).
View in article
Li, Y.L., Konhauser, K.O., Kappler, A., Hao, X.L. (2013b) Experimental low-grade alteration of biogenic magnetite indicates microbial involvement in generation of banded iron formations. Earth and Planetary Science Letters, 361, 229–237.
 Show in context
Show in context High pressure, high temperature (HPHT) experiments are now shedding light on microfossil taphonomy during geological sequestration, with a focus on thermal degradation of biogenic minerals (Li et al., 2013b) and biomineralised cells (Li et al., 2014).
View in article
Figure 1 [...] Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012; Li et al., 2013b, 2014; Picard et al., 2015a,b; Alleon et al., 2016, 2017; Miot et al., 2017).
View in article
Li, J., Bernard, S., Benzerara, K., Beyssac, O., Allard, T., Cosmidis, J., Moussou, J. (2014) Impact of biomineralization on the preservation of microorganisms during fossilization: An experimental perspective. Earth and Planetary Science Letters 400, 113–122.
 Show in context
Show in context High pressure, high temperature (HPHT) experiments are now shedding light on microfossil taphonomy during geological sequestration, with a focus on thermal degradation of biogenic minerals (Li et al., 2013b) and biomineralised cells (Li et al., 2014).
View in article
Figure 1 [...] Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012; Li et al., 2013b, 2014; Picard et al., 2015a,b; Alleon et al., 2016, 2017; Miot et al., 2017).
View in article
This limitation was also observed by Li et al. (2014) for experiments at similar temperatures (300 ºC, 600 ºC) in the absence of pressure.
View in article
Moreau, J.W., Sharp, T.G. (2004) A Transmission Electron Microscopy Study of Silica and Kerogen Biosignatures in ~1.9 Ga Gunflint Microfossils. Astrobiology 4, 196–210.
 Show in context
Show in context Chemically-precipitated sediments, particularly carbonate and silica, host microfossils spanning much of Earth history, including in association with early evidence for microbial life on Earth (Moreau and Sharp, 2004; Westall, 2008; Wacey et al., 2011; Djokic et al., 2017).
View in article
Displacement of kerogen during diagenetic coarsening of quartz has been identified as a mechanism to explain petrographic relationships in the microfossil-bearing Gunflint chert (Moreau and Sharp, 2004; Foucher and Westall, 2013).
View in article
Miot, J., Bernard, S., Bourreau, M., Guyot, F., Kish, A. (2017) Experimental maturation of Archaea encrusted by Fe-phosphates. Scientific Reports 7, 16984.
 Show in context
Show in context Figure 1 [...] Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012; Li et al., 2013b, 2014; Picard et al., 2015a,b; Alleon et al., 2016, 2017; Miot et al., 2017).
View in article
Orange, F., Disnar, J.R., Gautret, P., Westall, F., Bienvenu, N., Lottier, N., Prieur, D. (2012) Preservation and evolution of organic matter during experimental fossilisation of the hyperthermophilic archaea Methanocaldococcus jannaschii. Origins of Life and Evolution of Biospheres 42, 587–609.
 Show in context
Show in context Significant experimental effort has furthered our understanding of microbial biomineralisation (Westall et al., 1995; Orange et al., 2012, 2014; Li et al., 2013a and references therein; Gaboyer et al., 2017).
View in article
Orange, F., Dupont, S., Goff, O.L., Bienvenu, N., Disnar, J.R., Westall, F., Le Romancer, M., (2014) Experimental fossilization of the thermophilic Gram-positive Bacterium Geobacillus SP7A: a long duration preservation study. Geomicrobiology Journal, 31, 578–589.
 Show in context
Show in context Significant experimental effort has furthered our understanding of microbial biomineralisation (Westall et al., 1995; Orange et al., 2012, 2014; Li et al., 2013a and references therein; Gaboyer et al., 2017).
View in article
Papineau, D., DeGregorio, B., Sagar, J., Thorogate, R., Wang, J., Nittler, L., Kilcoyne, D.A., Marbach, H., Drost, M. Thornton, G. (2019) Fossil biomass preserved as graphitic carbon in a late Paleoproterozoic banded iron formation metamorphosed at more than 550 ºC. Journal of the Geological Society of London 176, 651-668.
 Show in context
Show in context As improving analytical techniques expand biosignature studies into increasingly metamorphosed terrains (Bernard et al., 2007, 2010; Galvez et al., 2012; Papineau et al., 2019), there is a need to understand the influence of higher PT regimes (Li et al., 2013a).
View in article
Picard, A., Kappler, A., Schmid, G., Quaroni, L., Obst, M. (2015a) Experimental diagenesis of organo-mineral structures formed by microaerophilic Fe(II)-oxidizing bacteria. Nature Communications 6, 6277 (2015), doi: 10.1038/ncomms7277.
 Show in context
Show in context Experiments have demonstrated the persistence of microbiological structures such as iron oxide organominerals up to 250 °C and 140 MPa (Picard et al., 2015a), and the preservation of lipids and polysaccharides within Fe-encrusted microbial cells at the same PT conditions (Picard et al., 2015b).
View in article
Figure 1 [...] Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012; Li et al., 2013b, 2014; Picard et al., 2015a,b; Alleon et al., 2016, 2017; Miot et al., 2017).
View in article
The transformation of Fe minerals to more stable, crystalline phases has also been shown to be conducive to the preservation of organic components (Ferris et al., 1988; Picard et al., 2015a).
View in article
Picard, A., Obst, M., Schmid, G., Zeitvogel, F., Kappler, A. (2015b) Limited influence of Si on the preservation of Fe mineral-encrusted microbial cells during experimental diagenesis Geobiology 14, 276–292.
 Show in context
Show in context Experiments have demonstrated the persistence of microbiological structures such as iron oxide organominerals up to 250 °C and 140 MPa (Picard et al., 2015a), and the preservation of lipids and polysaccharides within Fe-encrusted microbial cells at the same PT conditions (Picard et al., 2015b).
View in article
Figure 1 [...] Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012; Li et al., 2013b, 2014; Picard et al., 2015a,b; Alleon et al., 2016, 2017; Miot et al., 2017).
View in article
Pimenta, M.A., Dresselhaus, G., Dresselhaus, M.S., Cancado, L.G., Jorio, A., Saito, R. (2007) Studying disorder in graphite-based systems by Raman spectroscopy. Physical Chemistry Chemical Physics 9, 1276–1291.
 Show in context
Show in context Raman spectra of experimental sample surfaces exhibit peaks for carbonate (1100 cm-1), and D1 (1360 cm-1) and G (1610 cm-1) carbon bands (Fig. 2; Pimenta et al., 2007).
View in article
Schiffbauer, J. D., Wallace, A.F., Hunter, J.L., Kowalewski, M., Bodnar, R.J., Xiao, S. (2012) Thermally‐induced structural and chemical alteration of organic‐walled microfossils: an experimental approach to understanding fossil preservation in metasediments. Geobiology 10, 402–423.
 Show in context
Show in context Figure 1 [...] Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012; Li et al., 2013b, 2014; Picard et al., 2015a,b; Alleon et al., 2016, 2017; Miot et al., 2017).
View in article
Starke, V., Kirshtein, J., Fogel, M.L., Steele, A. (2013) Microbial community composition and endolith colonization at an Arctic thermal spring are driven by calcite precipitation. Environmental Microbiology Reports 5, 648-659, doi: 10.1111/1758-2229.12063.
 Show in context
Show in context Porous carbonate precipitates containing green, orange, and grey chasmolithic microbial communities (Fig. 1c) were collected from a CO2 thermal spring (Jotun Spring; Banks et al., 1998) in Spitzbergen, Svalbard, during the Arctic Mars Analog Svalbard Expeditions from 2006-2011 (Starke et al., 2013).
View in article
Toporski, J.K., Steele, A., Westall, F., Thomas-Keprta, K.L., McKay, D.S. (2002) The simulated silicification of bacteria - new clues to the modes and timing of bacterial preservation and implications for the search for extraterrestrial microfossils, Astrobiology 2, 1–26.
 Show in context
Show in context This has implications for the longevity of biosignatures, whereby the quartz fraction resists weathering or secondary aqueous alteration, protecting captured OM (Toporski et al., 2002).
View in article
Wacey, D., Kilburn, M.R., Saunders, M., Cliff, J., Brasier, M.D. (2011) Microfossils of sulphur-metabolizing cells in 3.4-billion-year-old rocks of Western Australia. Nature Geoscience 4, 698–702.
 Show in context
Show in context Chemically-precipitated sediments, particularly carbonate and silica, host microfossils spanning much of Earth history, including in association with early evidence for microbial life on Earth (Moreau and Sharp, 2004; Westall, 2008; Wacey et al., 2011; Djokic et al., 2017).
View in article
The petrographic relationships between microbially-derived kerogen and the inorganic matrix are important in establishing microfossil biogenicity (Brasier et al., 2005; Wacey et al., 2011; Foucher and Westall, 2013), in addition to understanding broader interations of microbe-mineral systems back into the early Archean (Westall et al., 2015).
View in article
Their preservation at the highest HPHT condition holds promise for establishing the biogenicity of similar features in filmentous microfossils (Wacey et al., 2011; Bailey et al., 2013) and distinguishing them from abiotic S microstructures (Cosmidis and Templeton, 2016).
View in article
Westall, F., Boni, L., Guerzoni, E. (1995) The experimental silicification of microorganisms. Palaeontology 38, 495–528.
 Show in context
Show in context Significant experimental effort has furthered our understanding of microbial biomineralisation (Westall et al., 1995; Orange et al., 2012, 2014; Li et al., 2013a and references therein; Gaboyer et al., 2017).
View in article
Westall, F. (2008) Morphological Biosignatures in Early Terrestrial and Extraterrestrial Materials. Space Science Reviews 135, 95–114.
 Show in context
Show in context Our knowledge of microbiological evolution is informed by organic, morphological, and geochemical biosignatures preserved within a rock record that is destroyed or modified over geological time (Westall, 2008).
View in article
Chemically-precipitated sediments, particularly carbonate and silica, host microfossils spanning much of Earth history, including in association with early evidence for microbial life on Earth (Moreau and Sharp, 2004; Westall, 2008; Wacey et al., 2011; Djokic et al., 2017).
View in article
Westall, F., Campbell, K.A., Bréhéret, J.G., Foucher, F., Gautret, P., Hubert, A., Sorieul, S., Grassineau, N., Guido, D.M. (2015) Archean (3.33 Ga) microbe-sediment systems were diverse and flourished in a hydrothermal context. Geology 43, 615–618.
 Show in context
Show in context The petrographic relationships between microbially-derived kerogen and the inorganic matrix are important in establishing microfossil biogenicity (Brasier et al., 2005; Wacey et al., 2011; Foucher and Westall, 2013), in addition to understanding broader interations of microbe-mineral systems back into the early Archean (Westall et al., 2015).
View in article
top
Supplementary Information
The Supplementary Information includes:
- Methods
- Figures S-1 to S-5
Download the Supplementary Information (PDF).
Figures and Tables

Figure 1 Starting material for experiments showing (a) Six high pressure, high temperature (HPHT) experimental conditions investigated and their corresponding metamorphic grades spanning lower and upper blueschist (BS), greenschist (GS) and amphibolite (Amph.). Grey depicts HPHT conditions investigated previously (Schiffbauer et al., 2012
Schiffbauer, J. D., Wallace, A.F., Hunter, J.L., Kowalewski, M., Bodnar, R.J., Xiao, S. (2012) Thermally‐induced structural and chemical alteration of organic‐walled microfossils: an experimental approach to understanding fossil preservation in metasediments. Geobiology 10, 402–423.
; Li et al., 2013bLi, Y.L., Konhauser, K.O., Kappler, A., Hao, X.L. (2013b) Experimental low-grade alteration of biogenic magnetite indicates microbial involvement in generation of banded iron formations. Earth and Planetary Science Letters, 361, 229–237.
, 2014Li, J., Bernard, S., Benzerara, K., Beyssac, O., Allard, T., Cosmidis, J., Moussou, J. (2014) Impact of biomineralization on the preservation of microorganisms during fossilization: An experimental perspective. Earth and Planetary Science Letters 400, 113–122.
; Picard et al., 2015aPicard, A., Kappler, A., Schmid, G., Quaroni, L., Obst, M. (2015a) Experimental diagenesis of organo-mineral structures formed by microaerophilic Fe(II)-oxidizing bacteria. Nature Communications 6, 6277 (2015), doi: 10.1038/ncomms7277.
,bPicard, A., Obst, M., Schmid, G., Zeitvogel, F., Kappler, A. (2015b) Limited influence of Si on the preservation of Fe mineral-encrusted microbial cells during experimental diagenesis Geobiology 14, 276–292.
; Alleon et al., 2016Alleon, J., Bernard, S., Le Guillou, C., Daval, D., Skouri-Panet, F., Pont, S., Delbes, L., Robert, F. (2016) Early entombment within silica minimizes the molecular degradation of microorganisms during advanced diagenesis. Chemical Geology 437, 98–108.
, 2017Alleon, J., Bernard, S., Le Guillou, C., Daval, D., Skouri-Panet, F., Kuga, M., Robert, F. (2017) Organic molecular heterogeneities can withstand diagenesis. Scientific Reports 7, 1508.
; Miot et al., 2017Miot, J., Bernard, S., Bourreau, M., Guyot, F., Kish, A. (2017) Experimental maturation of Archaea encrusted by Fe-phosphates. Scientific Reports 7, 16984.
). (b) Si-rich microbial filamentous material and coccoidal cell structures are observed under SEM in the the starting material, shown in (c) by a view ~ 1 cm across; forming Si-rich biofilms around calcite grains (Fig. S-1a,b).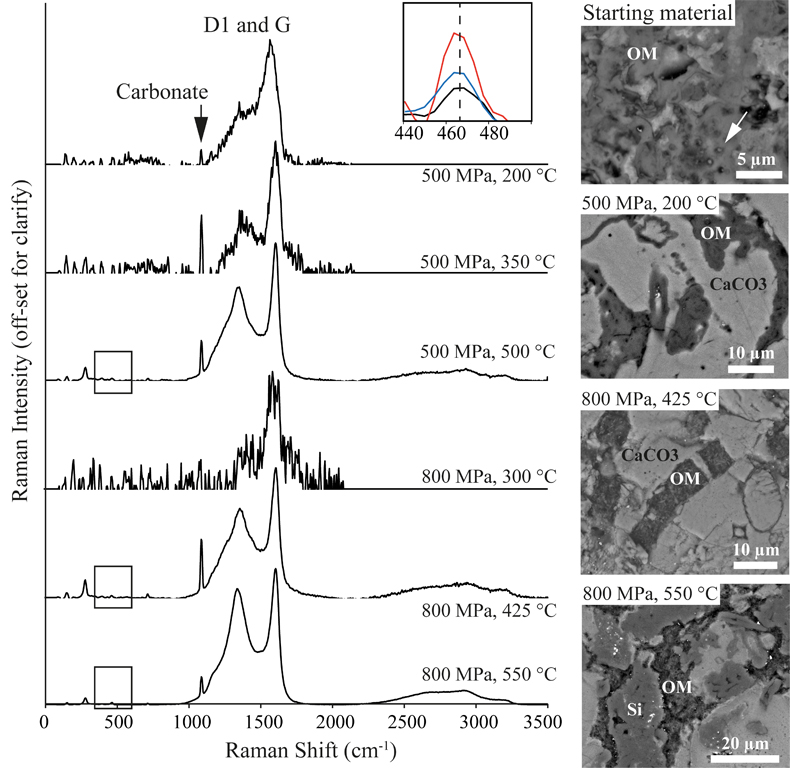
Figure 2 Evolution of D1 (~1360 cm-1) and G (~1610 cm-1) Raman peaks from experimental samples, and the increasing 467 cm-1 quartz peak at 425, 500, and 550 °C (box inset); and corresponding SEM images showing the textural evolution of OM mineralisation within the carbonate (CaCO3) matrix and siliceous (Si) phases Individual cells in the amorphous organic matrix can be seen in the starting material (arrows). Unprocessed Raman spectra are given in Figure S-3.

Figure 3 BSE+EDS elemental maps or plain BSE image (left), where red = Ca and green = Si, and corresponding Raman maps (right) of the same region, where fuchsia = calcite (1006 cm-1 band), orange/yellow = quartz (465 cm-1 band), green = carbon (combined D1 and G bands), light pink = resin, black = masked fluorescence. (a) Starting material; (b) 800 MPa, 300 °C; (c) 500 MPa, 200 °C; (d) 800 MPa, 425 °C; (e) 500 MPa, 350 °C; (f) 800 MPa, 550 °C; (g) 500 MPa, 500 °C; (h) 800 MPa, 550 °C. EDS spectra are provided in Figure S-5.

Figure 4 (a – g) BSE+EDS elemental maps (red = Ca, green = Si, yellow = S) and SEM images of sulfur globules in (a, b) Starting material; (c) 800 MPa, 425 °C; (d, e) 800 MPa, 300 °C; (f) 800 MPa, 550 °C; and (g) 500 MPa, 500 °C; (h) BSE+EDS elemental map showing the co-location of Fe (Fe = purple) with S (S = yellow) within a silica-rich (Si = red) fabric (i) in the 800 MPa, 425 °C experiment.